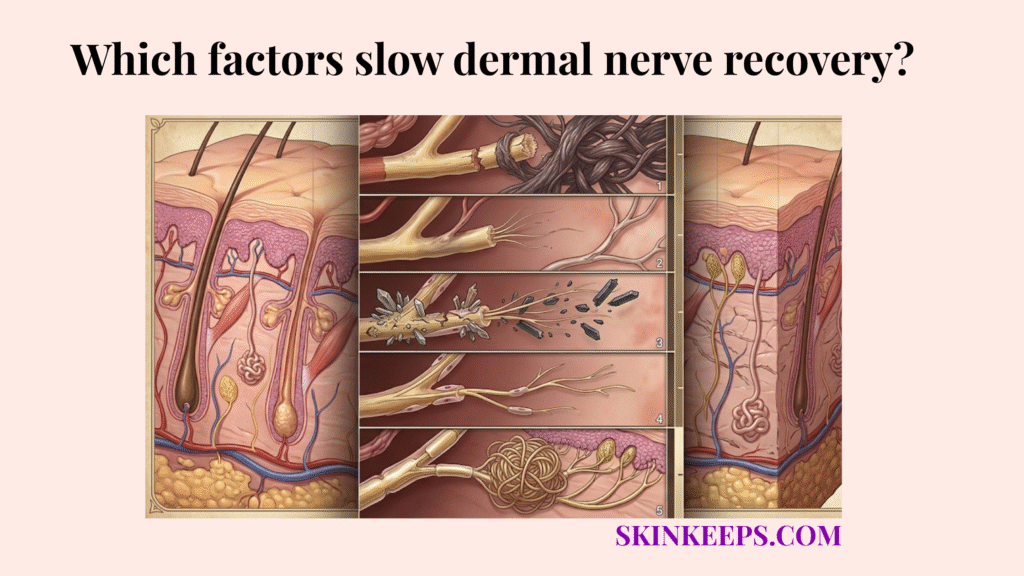
The main factors that slow dermal nerve recovery are poor glycemic control, reduced perfusion, chronic inflammation, fibrosis or scarring, metabolic deficiency, and age-related repair slowdown, because these conditions interfere with the nerve’s need for a clean path, strong blood supply, and steady metabolic support during regrowth.
Sensory return can stay delayed even when the skin surface looks completely healed because closing the physical skin layer is a rapid structural patch job, whereas peripheral nerve regeneration is a highly delicate, microscopic rewiring process.
Understanding exactly which barriers block this process requires evaluating physical obstructions, metabolic starvation, chemical hostility, aging factors, and the specific clinical interventions that help restore a supportive environment.
What are the primary biological factors that slow dermal nerve recovery?
The primary biological factors that slow dermal nerve recovery are the obstacles that block the growth path physically, weaken the regrowth environment metabolically, or destabilize it chemically through chronic inflammatory stress.
Physical barriers such as fibrosis act as mechanical roadblocks that trap regenerating fibers. Metabolic barriers such as glycemic dysfunction starve the nerve of the energy required to extend forward. Chemical barriers such as unresolved inflammation create a toxic microenvironment that misdirects or halts the repair sequence entirely.
By categorizing these impediments into distinct classes, it becomes obvious why the biology of dermal nerve regeneration requires a pristine, highly supported tissue environment to succeed.
How does glycemic control act as one of the factors that slow dermal nerve recovery?
Poor glycemic control acts as one of the major factors that slow dermal nerve recovery because chronic hyperglycemia damages the microvascular and metabolic support systems that regenerating nerves depend on.
Advanced glycation end products accumulate in the tissue, creating intense metabolic stress that damages the nerve’s internal repair biology. This microvascular damage effectively starves the advancing growth cone of essential nutrients.
Consequently, regrowth slows or fails completely, severely worsening recovery quality in cases of established diabetic neuropathy.
How does reduced perfusion represent critical factors that slow dermal nerve recovery?
Reduced perfusion represents one of the most critical factors that slow dermal nerve recovery because regenerating nerves require oxygen-rich blood flow to maintain ATP production, growth-cone activity, and axonal transport.
Without adequate oxygen delivery, the tissue suffers from hypoxia, stalling the massive energy output required for structural nerve extension. Smoking and vascular disease reduce this vital circulation, creating a starved healing environment.
Because the regeneration rate of dermal nerve fibers is biologically constrained to roughly 1 mm per day, as noted by Althagafi and Nadi (2023), any reduction in ATP supply can cause the advancing tip to halt completely [StatPearls].
How do fibrosis and scarring serve as factors that slow dermal nerve recovery?
Fibrosis and scarring serve as factors that slow dermal nerve recovery because perineural scar tissue and adhesion can distort, compress, trap, or misdirect the regenerating pathway.
Fibrosis creates a severe mechanical obstruction that physical nerve sprouts cannot easily penetrate. The nerve may be biologically active and attempting to regrow, but perineural scarring traps the growth cone in a physical dead-end.
This leads to misdirected growth or painful neuroma formation instead of successful reinnervation of the original skin target.
How does chronic inflammation become one of the factors that slow dermal nerve recovery?
Chronic inflammation becomes one of the factors that slow dermal nerve recovery because regeneration requires inflammation to resolve into a supportive repair state rather than remain persistently destructive.
While acute early inflammation helps clear debris, an unresolved inflammatory burden keeps the tissue chemically hostile. This persistent chemical instability destabilizes the delicate cues needed for orderly regrowth.
As a result, axonal progression becomes chaotic and significantly less efficient, preventing the nerve from re-establishing normal sensory links.
How do age and environmental stressors serve as factors that slow dermal nerve recovery?
Age and environmental stressors serve as factors that slow dermal nerve recovery because older tissue shows reduced regenerative capacity, while chronic environmental burdens such as smoking or long-term tissue stress reduce vascular resilience and repair efficiency.
Age-related regenerative decline lowers the efficiency of Schwann cells and slows the overall metabolic repair process. Simultaneously, chronic external stress further burdens the tissue, creating a compounded delay in sensory return.
| Factor type | Main effect on nerve regrowth | Recovery consequence | Common visible result |
|---|---|---|---|
| Aging | Slower repair Schwann-cell behavior and lower regenerative efficiency | Longer recovery timeline | Persistent numbness or slower sensory return |
| Environmental stress | Higher vascular and tissue burden | Less efficient healing environment | More fragile or delayed recovery |
| Chronic cumulative stress | Lower tissue resilience | Higher risk of stalled regrowth | Incomplete sensory normalization |
How do optimized healing environments compare with environments where factors slow dermal nerve recovery?
Optimized healing environments compare favorably with obstructed ones because dermal nerves recover best when oxygen delivery, metabolic support, inflammation control, and pathway clarity are all preserved at the same time.
In an optimized environment, supportive perfusion and stable metabolism clear the way for accurate reinnervation. In contrast, a hostile tissue environment combines poor circulation with heavy fibrosis, actively preventing sensory recovery.
No single factor acts in isolation; the holistic tissue context dictates the ultimate success of the repair.
What clinical interventions mitigate the factors that slow dermal nerve recovery?
Clinical interventions mitigate the factors that slow dermal nerve recovery by improving metabolic support, reducing glycation-related stress, limiting fibrosis-related obstruction, and preserving a more favorable repair pathway.
These therapies focus on stabilizing the nerve-healing environment rather than acting as magical instantaneous cures. Exploring which factors slow dermal nerve recovery reveals exactly why structural support outlasts quick-fix promises.
How do Vitamin B12 and Benfotiamine help mitigate the factors that slow dermal nerve recovery?
Vitamin B12 and benfotiamine may help mitigate the factors that slow dermal nerve recovery by improving aspects of neuronal metabolism, myelin-related support, and glycation-linked metabolic stress, but they should be presented as supportive measures rather than guaranteed regeneration accelerators.
Vitamin B12 provides essential neurotropic support for myelin biology and cellular energy. Benfotiamine targets glycemic stress, making it particularly useful in mitigating the metabolic damage associated with chronic hyperglycemia.
While these supports improve repair conditions, they do not guarantee faster reinnervation.
How do scar-management and tissue mobilization help mitigate the factors that slow dermal nerve recovery?
Scar-management and tissue-mobilization strategies may help mitigate the factors that slow dermal nerve recovery by reducing adhesion-related restriction, improving tissue glide, and limiting some of the mechanical dead-ends created by fibrosis, although evidence for direct acceleration of true axonal regrowth is less definitive than the rationale for reducing scar-related obstruction.
Tissue mobilization physically addresses the mechanical barriers that trap regenerating nerves. By reducing perineural scarring and improving scar glide, these techniques help restore a less restrictive path for the advancing axon.
Clinical Intervention Framework
- Problem → the nerve-healing environment is metabolically weak or physically blocked.
- Implication → the growth cone slows, stalls, or misroutes.
- Solution → improve metabolic support, reduce fibrosis burden, and restore a less restrictive repair pathway.
What are the key summary facts regarding factors that slow dermal nerve recovery?
The key summary facts are that the biggest factors that slow dermal nerve recovery are poor glycemic control, reduced perfusion, chronic inflammation, fibrosis, and age-related repair decline, because each of these barriers either blocks the growth path, starves the nerve metabolically, or destabilizes the repair environment.
Summary Checklist
What steps can you take to address the factors that slow dermal nerve recovery today?
The factors that slow dermal nerve recovery are addressed most effectively when patients stabilize metabolic stress, protect circulation, support nerve nutrition, and reassess abnormal recovery early instead of assuming that healed skin means healed sensation.
Final Execution Checklist
Quick Answers About Which Factors Slow Dermal Nerve Recovery
Which factors slow dermal nerve recovery the most?
The factors that slow dermal nerve recovery the most are poor glycemic control, reduced perfusion, severe fibrosis, and chronic inflammation. These systemic and local barriers physically block the regrowth path or metabolically starve the advancing nerve.
Does diabetes slow dermal nerve recovery?
Yes. Glycemic dysfunction creates advanced glycation end products and microvascular damage, starving the nerve of oxygen and generating metabolic stress that severely stalls regenerative progress.
Why does poor circulation delay nerve regrowth?
Poor circulation delays nerve regrowth because the advancing growth cone requires massive amounts of ATP. Reduced perfusion causes tissue hypoxia, shutting down the energy supply necessary for structural nerve extension.
Can scar tissue block nerve recovery?
Yes. Fibrosis and perineural scarring act as rigid mechanical roadblocks. They compress the repair pathway, trapping the nerve and often forcing it into a painful, misdirected tangle known as a neuroma.
Does age reduce nerve repair efficiency?
Yes. Age-related regenerative decline slows down Schwann-cell response times and weakens overall metabolic efficiency, ensuring that older tissue takes significantly longer to achieve functional sensory return.
Do B12 or benfotiamine help nerve recovery?
Vitamin B12 and benfotiamine support the metabolic environment required for healthy repair. They help mitigate glycemic stress and support myelin biology, though they act as supportive tools rather than instant regenerative cures.
Can smoking slow sensory return?
Yes. Smoking creates a hostile tissue environment by drastically reducing microvascular perfusion and increasing oxidative stress, effectively starving the regenerating nerve of the oxygen it needs to heal.
What signs suggest nerve recovery has stalled?
Persistent numbness, unusual hypersensitivity, shooting pains, or a complete lack of sensory progress months after the visible skin surface has completely closed strongly suggest that nerve recovery has stalled.
Conclusion
In conclusion, factors slow dermal nerve recovery when they block the nerve’s path, starve its metabolism, or destabilize the repair environment, which is why delayed sensory return often reflects deeper biological roadblocks rather than surface-healing failure alone.
Whether obstructed by heavy scarring, starved by poor perfusion, overwhelmed by metabolic stress, or compromised by chronic inflammation and age, the delicate regrowth of a peripheral nerve demands a highly optimized microenvironment. By targeting the exact barrier impeding growth rather than waiting passively, we drastically improve the odds of meaningful sensory reconnection.