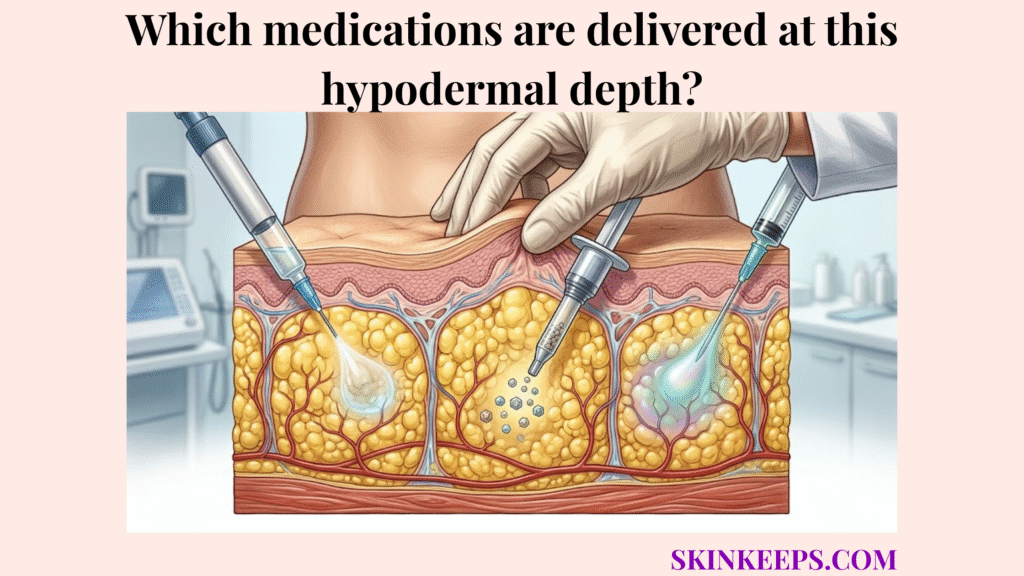
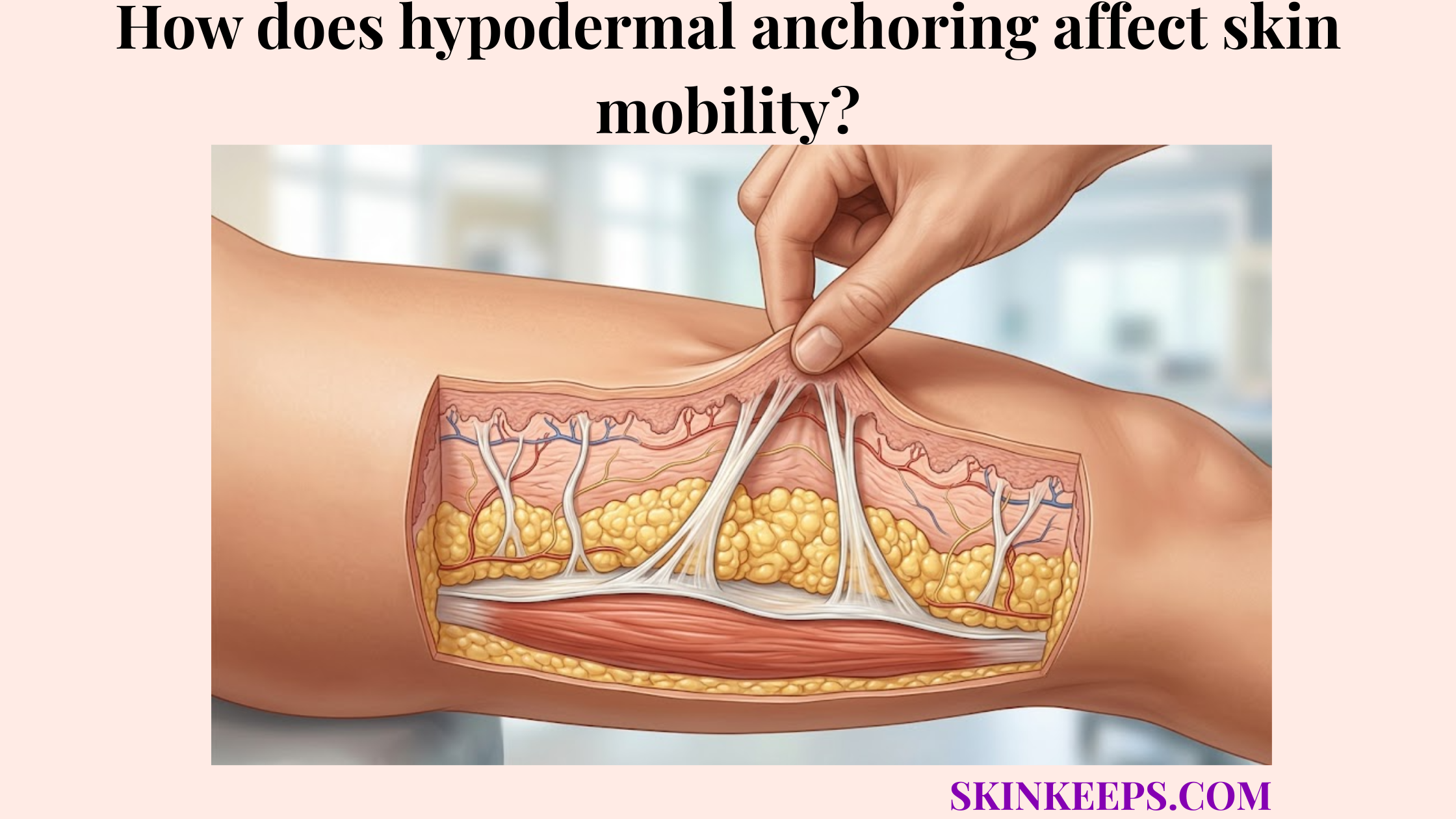
Medications delivered at the hypodermal depth include selected endocrine drugs, peptide therapies, biologics, fertility hormones, growth hormone, and anticoagulants that are specifically formulated and labeled for subcutaneous tissue administration. These medications use the hypodermis because its adipose tissue, extracellular matrix, capillaries, and lymphatic pathways can support controlled absorption when the product is designed for that route.
This guide explains what makes a drug compatible with the subcutaneous layer, why insulin and GLP-1 drugs commonly use it, how biologics and anticoagulants differ, and why some compounds must not enter subcutaneous fat. Readers should follow product labeling and clinician guidance because injection route, site, volume, device, and technique are medication-specific.
What characterizes the pharmacological profile of drugs approved for the subcutaneous layer?
The pharmacological profile of drugs approved for the subcutaneous layer is usually low-volume, tissue-compatible, non-vesicant, route-labeled, and designed for absorption through adipose tissue, capillaries, or lymphatic pathways. A compatible drug formulation ensures that the liquid can integrate safely into the fat layer without causing excessive damage. Because the hypodermis supports adipose storage, insulation, and structural anchoring, it can accommodate these specific fluids.
Understanding this subcutaneous injection profile separates safely formulated medications from those requiring deeper muscular or direct intravenous delivery. This profile ensures that the targeted subcutaneous tissue effectively manages the drug.
What volume and viscosity limits matter for subcutaneous medications?
Volume and viscosity limits matter for subcutaneous medications because the hypodermal extracellular matrix can only tolerate selected fluid volumes and formulation thickness before discomfort, leakage, delayed absorption, or local tissue pressure becomes more likely. Injection volume determines how much fluid the tissue can physically accept, while viscosity affects the force needed to push the medication through small needles.
A literature review reports that the maximum volume generally accepted for subcutaneous injection is around 1.5 mL, although volumes up to 3 mL may be tolerated in some contexts, so volume claims must remain medication-, site-, formulation-, and protocol-specific. [PMC]
What pH and isotonicity profile supports safe subcutaneous delivery?
A safe pH and isotonicity profile supports subcutaneous delivery when the formulation is compatible with local tissue fluid and does not create excessive chemical irritation in adipose tissue. When a formulation matches the body’s natural pH and isotonicity, the subcutaneous fat readily accepts the fluid. This tissue compatibility minimizes burning or stinging upon entry.
Avoiding highly irritating formulas prevents sterile inflammation or severe tissue injury. Therefore, maintaining a non-vesicant, tissue-compatible profile is essential for safe subcutaneous drug administration.
| Compatibility Factor | What It Means | Why It Matters |
|---|---|---|
| Low volume | Amount fits the intended site and protocol | Reduces tissue pressure and leakage risk |
| Appropriate viscosity | Formulation can pass through intended device | Reduces injection force and discomfort |
| Tissue-compatible pH | Formulation does not strongly irritate local tissue | Lowers local reaction risk |
| Appropriate osmolarity | Formulation does not strongly disrupt tissue fluid balance | Reduces irritation and pain |
| Non-vesicant profile | Drug is not expected to destroy tissue if correctly given | Protects fat and skin structures |
| Label-approved route | Product says SC/SQ or clinician directs SC use | Prevents route mismatch |
Why are endocrine hormones commonly delivered at the hypodermal depth?
Endocrine hormones are commonly delivered at the hypodermal depth because many injectable hormone formulations need gradual systemic absorption without being broken down in the digestive tract. The hypodermis provides an ideal environment for this controlled, tissue-based uptake. By forming a localized depot effect, these medications bypass immediate gastrointestinal destruction.
This gradual systemic entry allows these endocrine subcutaneous medications to mimic natural hormonal release over time. Clinical guidelines and product labels carefully dictate exactly how these agents should be administered.
Why are insulin formulations commonly injected into subcutaneous tissue?
Insulin formulations are commonly injected into subcutaneous tissue because many insulin products are designed to enter circulation from the fat layer at a formulation-specific rate rather than being delivered directly into blood or muscle. Both basal insulin and rapid-acting insulin rely on this consistent subcutaneous injection pathway. If these products accidentally enter skeletal muscle, they may absorb faster and increase the risk of unexpected hypoglycemia.
Therefore, precise depth control ensures the medication performs exactly as engineered. Following proper technique maintains the intended absorption profile and promotes safer blood glucose management.
Why are GLP-1 receptor agonists such as semaglutide and tirzepatide injected subcutaneously?
GLP-1 receptor agonists such as semaglutide and tirzepatide are injected subcutaneously because their injectable formulations are designed for gradual systemic exposure from subcutaneous tissue under product-specific dosing schedules. These GLP-1 receptor agonists act as peptide-based metabolic therapies that require a stable delivery route. The subcutaneous layer compatibility provides the necessary foundation for this sustained release.
Ozempic labeling describes semaglutide as an injection for subcutaneous use with once-weekly administration, and Mounjaro labeling describes tirzepatide as an injection for subcutaneous use containing a once-weekly GIP/GLP-1 receptor agonist.
| Drug Type | Example | Why It Uses SC Route | Duration Language |
|---|---|---|---|
| Rapid-acting insulin | Insulin aspart/lispro/glulisine | Designed for subcutaneous uptake | Product-specific rapid profile |
| Basal insulin | Glargine/degludec/detemir | Designed for prolonged absorption | Product-specific long profile |
| GLP-1 receptor agonist | Semaglutide | SC formulation supports gradual exposure | Often weekly depending on product |
| GIP/GLP-1 agonist | Tirzepatide | SC formulation supports systemic exposure | Once weekly in Mounjaro label |
| Fertility hormones | FSH/LH products | Some are prescribed SC under protocols | Cycle-specific |
| Growth hormone | Somatropin products | Many are SC under prescribed regimens | Product-specific |
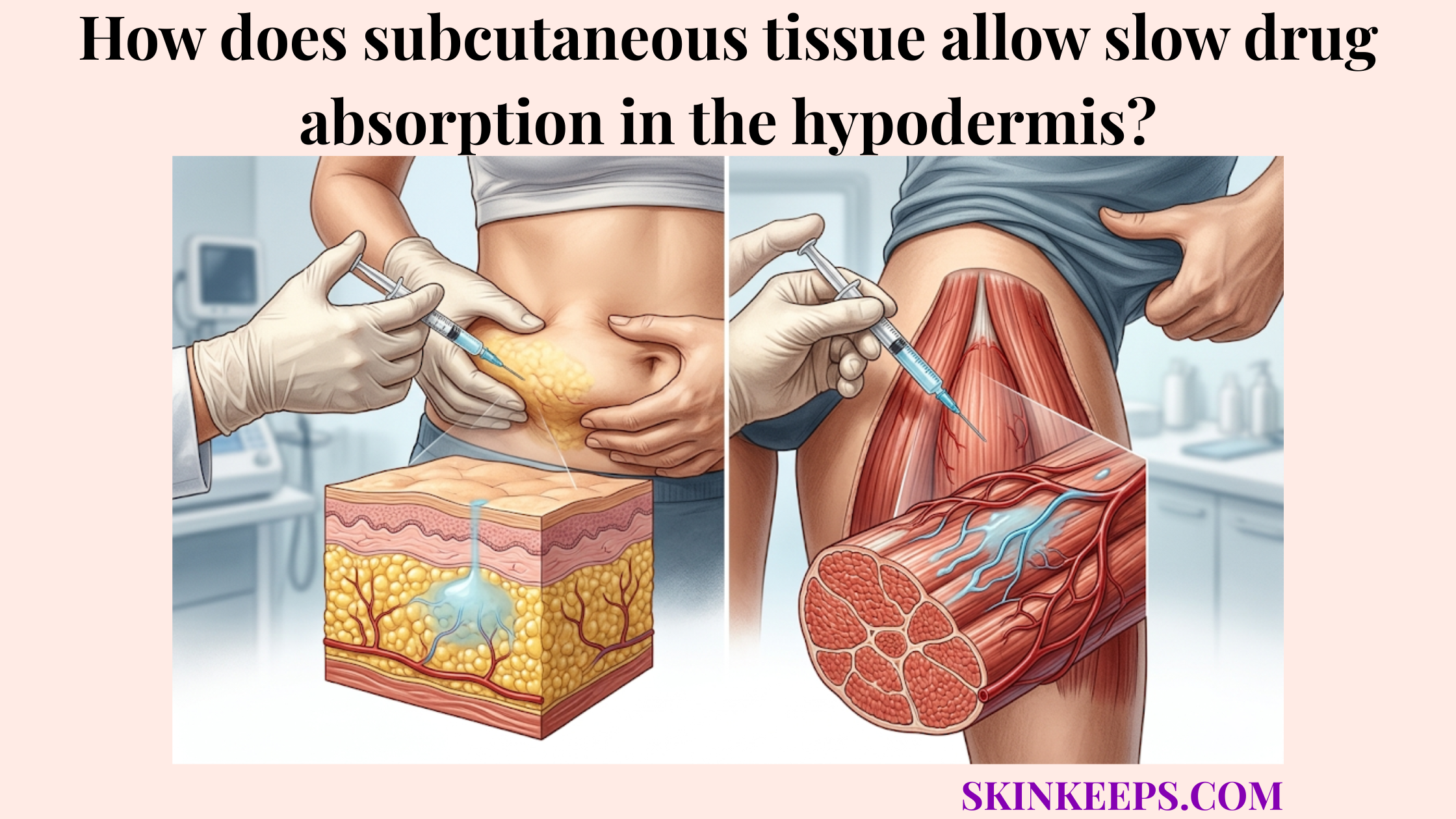
How does the deep fatty layer accommodate large-molecule biologic therapies?
The deep fatty layer accommodates large-molecule biologic therapies by allowing protein drugs to move through interstitial fluid and lymphatic pathways before reaching systemic circulation. Because blood flow supports hypodermal heat retention and moderate perfusion, the tissue relies heavily on these alternative pathways. Capillary uptake serves smaller molecules, while lymphatic absorption often contributes more heavily to transporting bulkier therapeutic proteins.
This specialized convective transport ensures that sensitive biologics eventually reach the bloodstream safely. Consequently, this route supports treatments that cannot simply diffuse through tiny capillary walls.
How do monoclonal antibodies use subcutaneous and lymphatic absorption?
Monoclonal antibodies use subcutaneous and lymphatic absorption by moving from the injection depot through interstitial spaces, with lymphatic transport often contributing more than direct capillary uptake for large protein molecules. These complex monoclonal antibodies navigate the fat lobules and slowly enter the highly permeable lymphatic vessels. This intricate journey creates the characteristically slow absorption profile seen with many immunotherapies.
Humira labeling identifies adalimumab as an injection for subcutaneous use, and Dupixent labeling identifies dupilumab as an injection for subcutaneous use; use these as examples of SC biologics rather than proof that every monoclonal antibody is SC.
How do fertility hormones and growth hormone use hypodermal delivery?
Fertility hormones and human growth hormone use hypodermal delivery by entering systemic circulation from subcutaneous tissue in prescribed, product-specific regimens designed to mimic or support hormonal signaling. Fertility hormones and human growth hormone depend on this stable tissue environment to achieve intended systemic concentrations. The soft adipose tissue accepts these specialized peptides and facilitates their gradual release.
Because formulations vary greatly, the exact timing and technique are strictly clinician-directed. Patients must adhere to their specific medical protocols to ensure safety and effectiveness.
| Drug Category | Example | Main Transport Concept | Safety Language |
|---|---|---|---|
| Monoclonal antibody | Adalimumab | Interstitial movement + lymphatic contribution | Product-label route only |
| Type 2 inflammation biologic | Dupilumab | SC depot with gradual systemic absorption | Follow label and prescriber |
| Fertility hormone | FSH/LH products | SC uptake under fertility protocol | Clinician-timed |
| Growth hormone | Somatropin products | SC systemic absorption | Product-specific |
| Other biologics | Varies | Size/formulation-dependent absorption | Never assume route |
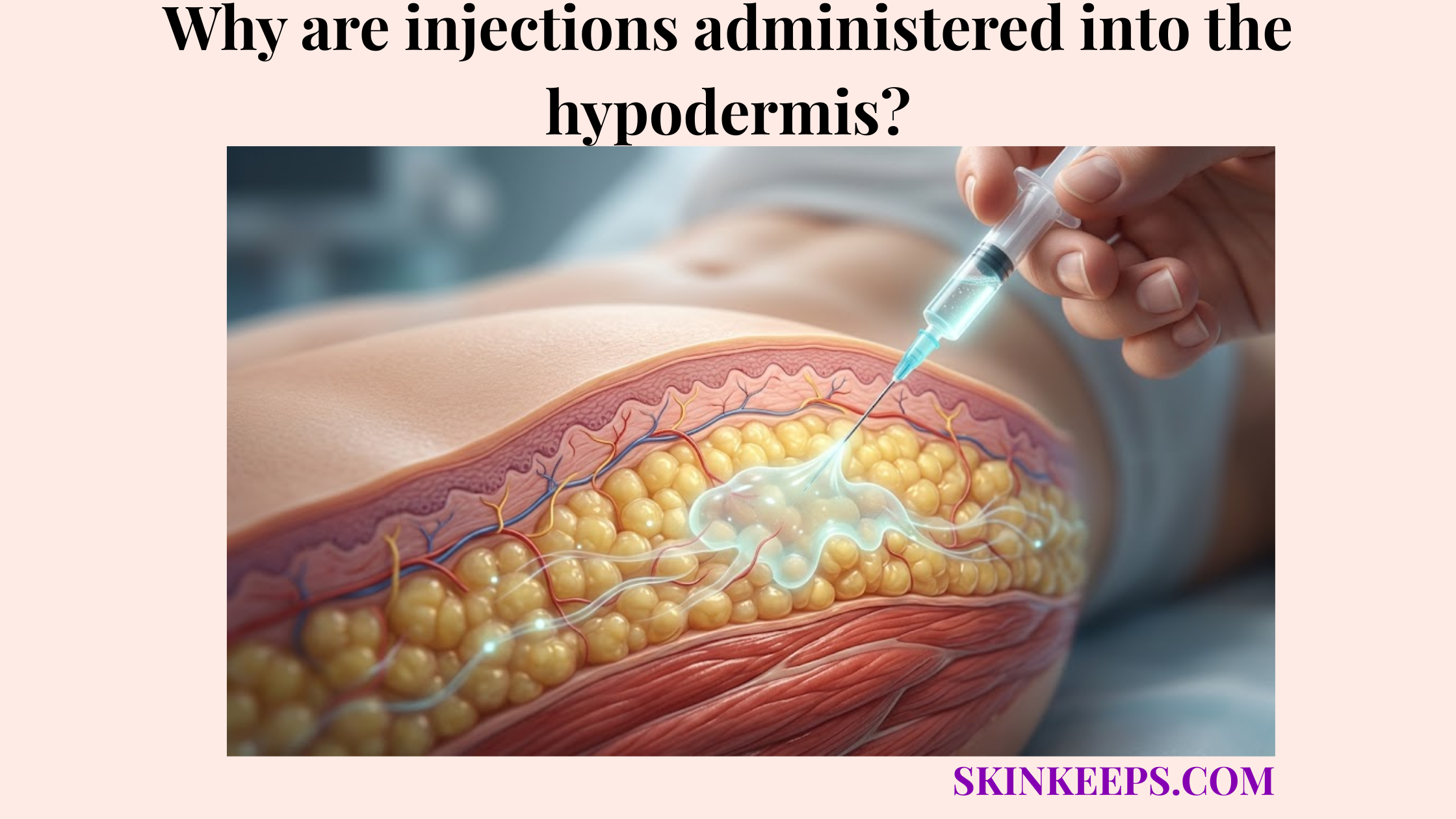
Why is deep subcutaneous administration used for specific anticoagulants?
Deep subcutaneous administration is used for specific anticoagulants because some heparin-based formulations are designed for predictable absorption from fatty tissue while avoiding direct injection into muscle. By targeting the fat layer, heparin formulations gain a steady, reliable entry into the bloodstream. Because fibrous septa connect layers in the hypodermis, the structure supports this necessary containment.
While this route manages systemic clotting risks, it also introduces local bruising or bleeding considerations. Therefore, these medications require strict adherence to clinical injection protocols.
Why are low-molecular-weight heparins often injected into abdominal subcutaneous tissue?
Low-molecular-weight heparins are often injected into abdominal subcutaneous tissue because products such as enoxaparin are formulated and taught for subcutaneous absorption under anticoagulation protocols. The abdominal wall provides a highly accessible, fat-rich area ideal for this specific low-molecular-weight heparin delivery. Selecting this site helps standardize the medication’s absorption profile across different patients.
UConn Health’s enoxaparin guide instructs injection into the right or left abdomen at least 2 inches from the belly button, while Sanofi’s Clexane guide instructs abdominal injection at least 5 cm from the umbilicus and advises not rubbing the site after administration. [UConn Health] and [Sanofi]
Why should anticoagulant injection technique follow the product label exactly?
Anticoagulant injection technique should follow the product label exactly because route, site, depth, and post-injection handling can affect bruising, bleeding risk, and medication delivery. For example, vigorously rubbing an enoxaparin injection site may exacerbate local hemorrhaging or cause significant hematomas. Such seemingly minor technique errors can dramatically compromise patient safety.
Anyone experiencing unusual bleeding, dark urine, or severe bruising should promptly consult their clinician. Practicing an exact anticoagulant subcutaneous protocol minimizes these preventable risks.
Which pharmaceutical compounds must not be injected into the hypodermal fat matrix unless specifically labeled for that route?
Pharmaceutical compounds that must not be injected into the hypodermal fat matrix unless specifically labeled for that route include vesicants, highly irritating solutions, concentrated electrolytes, inappropriate vasopressor formulations, and medications intended only for intramuscular or intravenous administration. These harsh chemicals can overwhelm the delicate subcutaneous fat architecture if accidentally introduced. Recognizing which drugs are strictly contraindicated for this route prevents severe tissue injuries.
Mismatching a potent medication with the incorrect tissue layer transforms a standard procedure into a potential clinical emergency. Healthcare providers rely on strict label verification to prevent these dangerous errors.
Which vasopressor or epinephrine formulations require special route caution?
Vasopressor and epinephrine formulations that require special route caution include IV vasopressor infusions such as norepinephrine and emergency epinephrine products whose route must follow the product label and clinical context. Intravenous vasopressors like norepinephrine can cause severe local ischemia if they accidentally leak into the fat. However, this differs entirely from a standard epinephrine auto-injector designed for specific emergency routes.
Norepinephrine extravasation is associated with subcutaneous tissue ischemia risk, while current EpiPen labeling says the auto-injector may be administered intramuscularly or subcutaneously into the anterolateral thigh; therefore, the writer must not say all epinephrine must never enter subcutaneous tissue.
Which irritating antibiotics and electrolytes are unsafe in subcutaneous tissue unless specifically directed?
Irritating antibiotics and electrolytes that are unsafe in subcutaneous tissue unless specifically directed include formulations such as IV vancomycin and concentrated potassium chloride when they extravasate or enter tissue outside the intended route. These powerful intravenous solutions act as dangerous vesicants if they pool in the hypodermis. When an extravasation occurs, the resulting chemical irritation severely compromises local blood vessels and soft tissue.
Vancomycin extravasation has been reported with local skin necrosis in case literature, and potassium chloride labeling lists extravasation-associated reactions including skin necrosis, soft tissue necrosis, and vascular injury; these examples show why route mismatch requires urgent clinical handling.
| Compound / Category | Intended Route Context | Problem if Wrong Tissue Is Hit | Safe Writer Language |
|---|---|---|---|
| Norepinephrine infusion | IV vasopressor | Extravasation can cause ischemic tissue injury | Urgent clinical assessment |
| EpiPen / epinephrine auto-injector | Label allows IM or SC thigh use | Do not confuse with IV vasopressor extravasation | Follow device label |
| Concentrated potassium chloride | IV with strict controls | Extravasation can cause severe tissue injury | Route mismatch is dangerous |
| Vancomycin IV | IV infusion | Extravasation can injure tissue | Case-reported necrosis |
| Adjuvanted vaccines | Usually IM | SC/ID route can increase local reactions | Follow vaccine guidance |
| IM-only depot drugs | Deep IM | SC placement may cause pain or erratic absorption | Follow label |
What clinical complications occur when intramuscular formulations enter the subcutaneous space?
Clinical complications from intramuscular formulations entering the subcutaneous space can include delayed absorption, local irritation, nodules, granuloma formation, reduced intended effect, or unpredictable medication release. Because skeletal muscle and the hypodermal depth possess entirely different vascular profiles, swapping their intended uses disrupts pharmacokinetics. An intramuscular formulation error forces the fat layer to manage substances it cannot efficiently process.
This mismatch frequently provokes an immune response, creating sterile inflammation or hard nodules beneath the skin surface. Adhering to the mandated injection depth averts these adverse reactions.
What happens when adjuvanted vaccines enter subcutaneous tissue instead of muscle?
When adjuvanted vaccines enter subcutaneous tissue instead of muscle, local irritation, induration, discoloration, inflammation, granuloma formation, or reduced effectiveness may become more likely depending on the vaccine and route error. Adjuvanted vaccines are typically formulated specifically for the deep vascularity of muscle tissue to optimize the immune response. Injecting them too shallowly traps the irritating components in the sensitive fat layer.
CDC vaccine-administration guidance states that non-live vaccines containing an adjuvant should be injected into muscle to decrease local adverse events, and subcutaneous or intradermal administration can cause local irritation, induration, skin discoloration, inflammation, and granuloma formation.
What happens when oil-based depot intramuscular medications are injected into fat?
When oil-based depot intramuscular medications are injected into fat, absorption may become delayed, painful, or unpredictable because the formulation was designed for deep muscle placement rather than subcutaneous tissue. These thick suspensions require robust muscular blood flow to dissolve appropriately. Leaving an oil-based depot formulation stranded in the hypodermis may result in a highly irritated, poorly absorbed sterile abscess.
Haldol decanoate labeling instructs deep intramuscular injection by a healthcare professional every 4 weeks, recommends a 21-gauge needle, and states the maximum volume per injection site should not exceed 3 mL in that IM-label context. [FDA]
| Intended Route | Wrong Placement | Main Risk | Example |
|---|---|---|---|
| Subcutaneous | Too deep into muscle | Faster absorption for some drugs | Insulin variability |
| Intramuscular | Too shallow into fat | Delayed or erratic absorption | IM depot medication |
| Adjuvanted IM vaccine | SC/ID tissue | Local irritation or granuloma | Some non-live vaccines |
| IV vesicant/irritant | Tissue extravasation | Local injury, ischemia, necrosis | Norepinephrine, KCl, vancomycin cases |
| Auto-injector | Wrong body site | Poor delivery or injury | Follow device label |
What is the pre-administration clinical checklist for verifying subcutaneous drug compatibility?
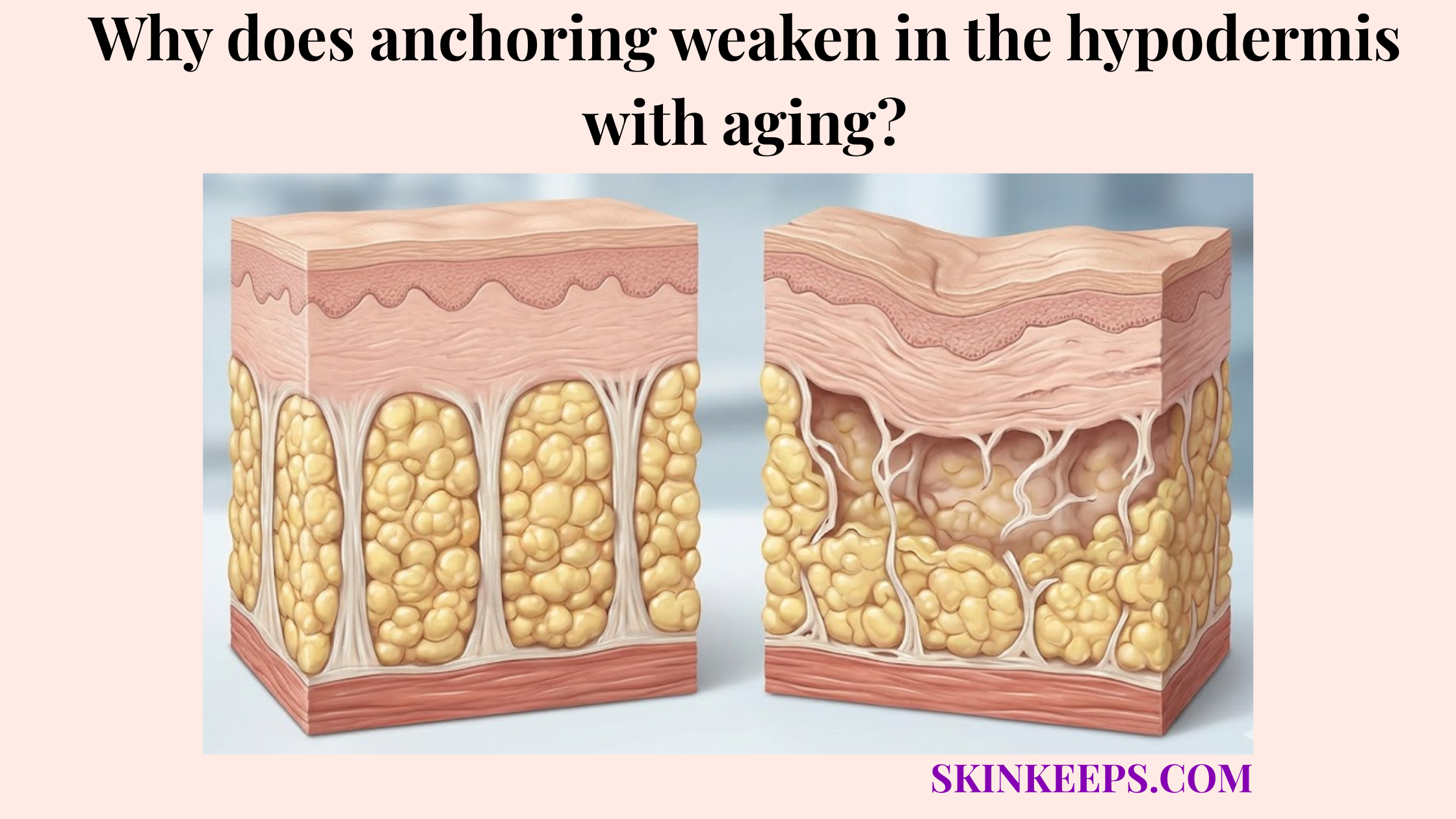
The pre-administration clinical checklist for verifying subcutaneous drug compatibility is to confirm the medication label, prescribed route, formulation, volume, site, device, contraindications, and patient-specific tissue conditions before administration. Executing a thorough medication route verification protocol guards against painful and potentially dangerous depth errors. Because hypodermal fat decreases with age, assessing individual patient anatomy remains equally critical.
Consistent pre-administration checks ensure the drug matches the tissue environment perfectly. This standard safeguards both efficacy and patient comfort during every procedure.
What should the physical profile check include?
The physical profile check should include injection volume, viscosity, formulation clarity, route label, device compatibility, and whether the tissue site can safely tolerate the medication. The clinician or patient must confirm that the target tissue is free of existing trauma, infection, or lipohypertrophy. Verifying these physical factors ensures the site can effectively absorb the intended dosage.
A literature review reports that the maximum volume generally accepted for subcutaneous injection is around 1.5 mL, although volumes up to 3 mL may be tolerated in some contexts, so volume claims must remain medication-, site-, formulation-, and protocol-specific. [PMC]
What should the pharmacological label check include?
The pharmacological label check should include the exact medication name, concentration, prescribed route, formulation type, route abbreviation, expiration status, contraindications, and whether the product exists in another route-specific version. Explicit labels like “For Subcutaneous Use” or “SC” confirm that the compound is chemically suited for the hypodermis. Missing this critical route-specific label check can lead to serious administration errors.
Whenever route abbreviations appear ambiguous, consulting a pharmacist or prescriber immediately clarifies the intended delivery path. Safety always supersedes assumptions when dealing with injectable therapeutics.
Hypodermal Medication Verification & Safety Checklist
FAQs About Medications Delivered at the Hypodermal Depth
Which medications are delivered at the hypodermal depth?
Medications delivered at the hypodermal depth include selected insulin products, GLP-1 receptor agonists, biologics, monoclonal antibodies, fertility hormones, growth hormone, and anticoagulants such as enoxaparin. Each product must be labeled or prescribed for subcutaneous use.
What makes a drug suitable for subcutaneous delivery?
A drug is suitable for subcutaneous delivery when its formulation, volume, viscosity, pH, tissue tolerance, absorption profile, and route labeling match the subcutaneous layer. Suitability is product-specific, so injectability alone does not prove SC compatibility.
Why are insulin and GLP-1 drugs often subcutaneous?
Insulin and many GLP-1 receptor agonists are often subcutaneous because their injectable formulations are designed for absorption through adipose tissue rather than direct bloodstream delivery. Their timing and effect depend on product design, site, and technique.
How do biologics absorb from subcutaneous tissue?
Biologics absorb from subcutaneous tissue by moving through interstitial fluid and, for many large proteins, partly through lymphatic pathways before reaching systemic circulation. This process is slower and more formulation-dependent than direct vascular injection.
Are epinephrine injections always forbidden in subcutaneous tissue?
No. Epinephrine route depends on product and context. Current EpiPen labeling allows intramuscular or subcutaneous injection into the anterolateral thigh, while IV vasopressor extravasation is a separate tissue-injury concern.
Why should some vaccines not be injected into fat?
Some adjuvanted vaccines should be injected into muscle because subcutaneous or intradermal administration can increase local irritation, induration, discoloration, inflammation, and granuloma formation. Route must follow vaccine guidance and manufacturer instructions.
What is the safest way to verify subcutaneous compatibility?
The safest verification method is to check the prescription, product label, route wording, formulation, device instructions, tissue site, and contraindications before administration. If route wording is unclear, a pharmacist, prescriber, or nurse should confirm compatibility before injection.
Conclusion
Medications delivered at the hypodermal depth are limited to products specifically formulated, labeled, or prescribed for subcutaneous tissue absorption. These include selected endocrine drugs, GLP-1 therapies, biologics, fertility or growth hormones, and anticoagulants, while vesicants, irritating IV drugs, and IM-only depot formulations require different route handling.
At SkinKeeps, we explain hypodermal anatomy through evidence-based dermatology and pharmacology. Understanding medication compatibility helps readers respect why route verification, product labeling, and clinician guidance matter before any drug enters the subcutaneous layer.