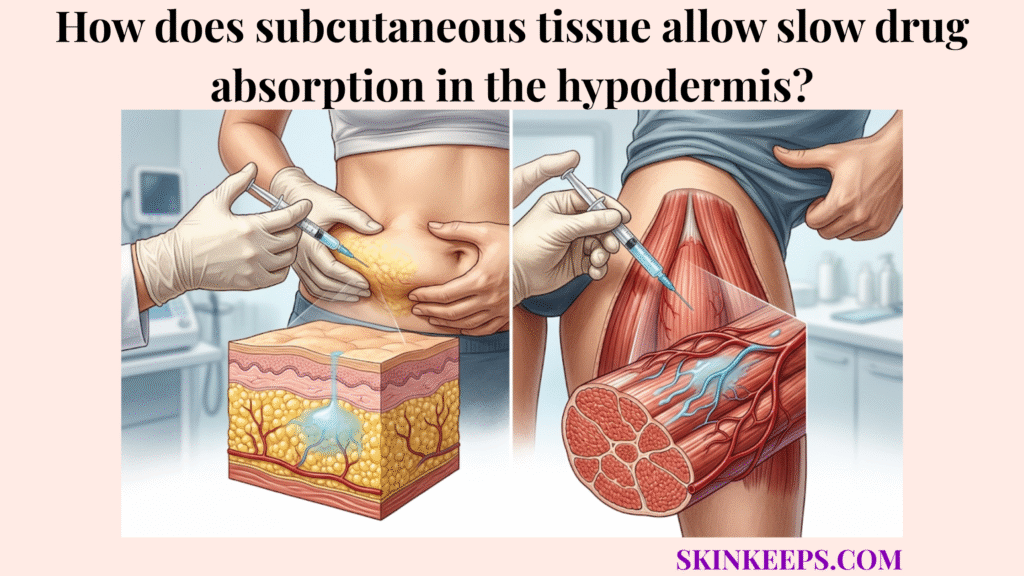
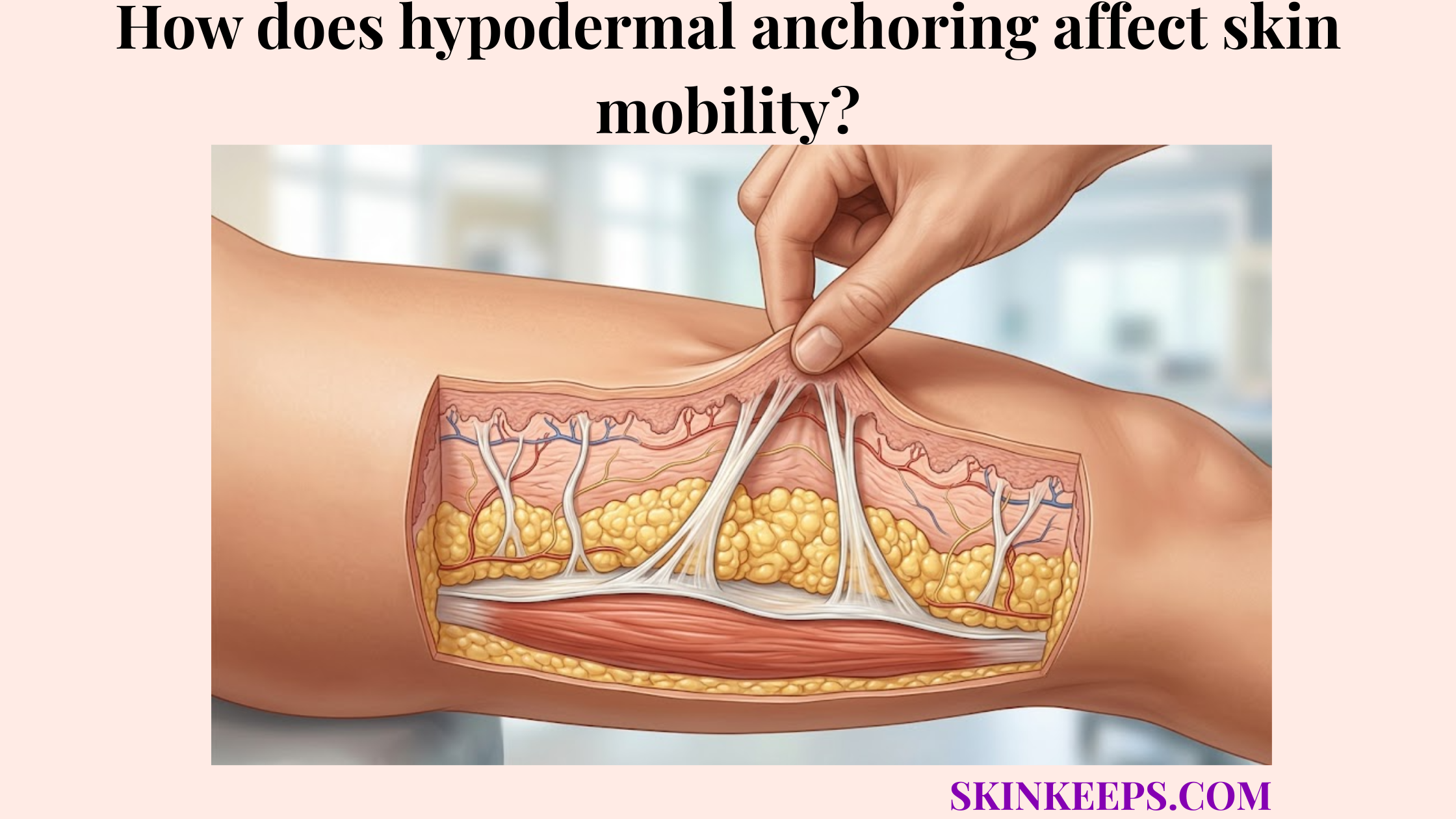
Subcutaneous tissue allows slow drug absorption in the hypodermis by making injected medication pass step-by-step through the hypodermal fat matrix, interstitial fluid, extracellular matrix, local capillaries, and sometimes lymphatic vessels before it reaches systemic circulation.
This guide explains how tissue structure, local blood flow, molecular size, depot formulations, edema, heat, exercise, massage, and injection-site condition can change the speed of subcutaneous absorption. Readers should follow medication labeling and clinician guidance because absorption is medication-specific, site-dependent, and technique-dependent.
What specific anatomical features of the hypodermal fat matrix govern medication uptake?
The specific anatomical features of the hypodermal fat matrix that govern medication uptake are its adipose lobules, extracellular matrix, interstitial fluid spaces, local capillaries, and lymphatic pathways. The hypodermis supports adipose storage, insulation, and structural anchoring, creating a highly organized subcutaneous tissue environment.
Because fibrous septa connect layers in the hypodermis, the medication must navigate a structured, three-dimensional barrier before it can be effectively absorbed.
How does the extracellular matrix slow drug diffusion?
The extracellular matrix slows drug diffusion by creating a hydrated connective environment that medication must move through before it reaches capillaries or lymphatic vessels. This ECM is filled with collagen and hyaluronic acid, which act together to modulate the flow of interstitial fluid.
Consequently, this tissue architecture influences the overall diffusion speed and delays the systemic absorption rate.
How do hydrophilic and lipophilic properties affect movement through adipose tissue?
Hydrophilic and lipophilic drug properties affect movement through adipose tissue because water-soluble and fat-soluble molecules interact differently with interstitial fluid, cell membranes, and lipid-rich compartments. Hydrophilic molecules typically favor the aqueous interstitial spaces, while lipophilic molecules may partition differently within the adipocytes.
These molecular interactions dictate how efficiently a drug navigates the fat lobules before reaching systemic circulation.
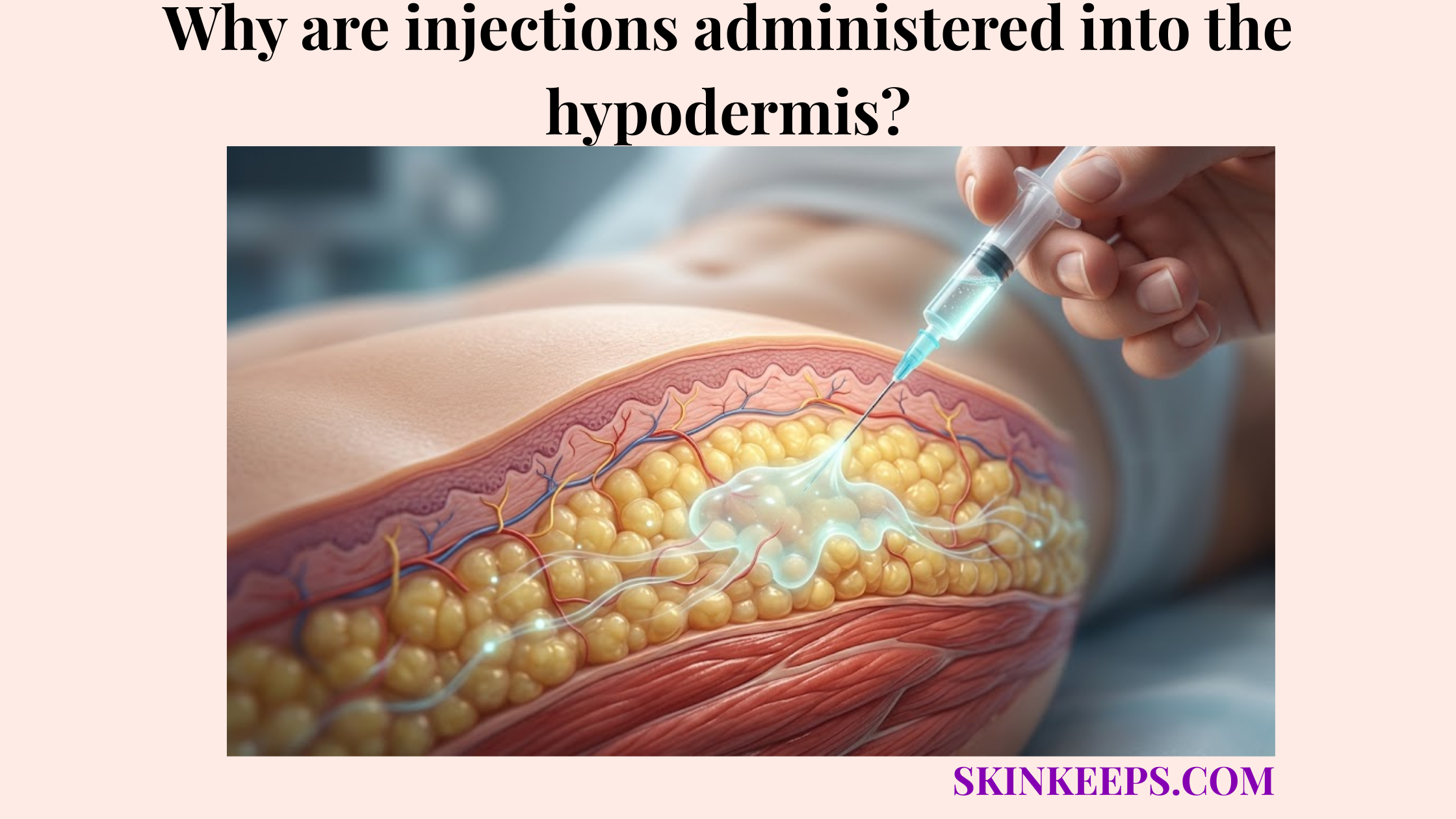
Why does the low capillary density of the subcutaneous layer support delayed systemic release?
The lower capillary density of the subcutaneous layer supports delayed systemic release because fewer immediate blood-entry pathways can make medication uptake slower than in more highly perfused tissues. Because blood flow supports hypodermal heat retention, this layer is moderately vascularized rather than heavily perfused like muscle.
This perfusion-limited environment prevents an immediate systemic surge of the injected medication.
How does perfusion-limited absorption work in subcutaneous fat?
Perfusion-limited absorption works in subcutaneous fat when local blood flow controls how quickly medication leaves the tissue depot and enters systemic circulation. The capillary uptake relies entirely on the local vascular density to carry the drug away.
Therefore, limited on-ramps into the bloodstream force the medication to linger safely in the subcutaneous depot.
How does resting blood flow create a more gradual absorption pattern?
Resting blood flow creates a more gradual absorption pattern when medication remains in the subcutaneous depot and enters circulation through relatively slower local perfusion. This steady, low-velocity microcirculation carefully meters the drug out of the fat lobules over time.
This highly controlled process supports predictable pharmacokinetics for specialized treatments.
| Tissue Type | Relative Perfusion | Main Uptake Pathway | Absorption Meaning |
|---|---|---|---|
| Subcutaneous fat | Often lower than muscle | Local capillaries and lymphatics | Can support slower depot-style absorption |
| Skeletal muscle | Generally more perfused | Capillary-rich muscle tissue | Can absorb some drugs faster |
| Dermis | Superficial vascular and nerve-rich layer | Small-volume intradermal use | Not ideal for many SC-formulated drugs |
| Intravenous route | Direct bloodstream access | No tissue depot | Immediate systemic exposure |
How do molecular size and weight affect drug diffusion through the adipose tissue compartment?
Molecular size and weight affect drug diffusion through the adipose tissue compartment because small molecules, peptides, and large biologics use different combinations of capillary uptake, interstitial diffusion, and lymphatic transport. The molecular weight determines which transport pathway the drug predominantly follows.
This physical limitation dictates the drug’s eventual bioavailability and onset of action.
How do small molecules enter local capillaries?
Small molecules can enter local capillaries more readily because their size and physicochemical properties may allow diffusion from interstitial fluid into nearby blood vessels. Their compact structure permits them to pass through capillary walls with relative ease.
This process generally results in faster systemic circulation compared to larger, bulkier therapeutics.
Why do large proteins and biologics rely more on lymphatic transport?
Large proteins and biologics rely more on lymphatic transport because their molecular size can limit direct blood-capillary uptake and increase dependence on convective movement through interstitial and lymphatic pathways. These macromolecules, such as monoclonal antibodies, enter the more permeable lymphatic vessels to return to the bloodstream.
A Drug Delivery Today review reports that large proteins in the approximately 30–40 kDa range showed almost complete absorption through peripheral lymphatics in a sheep model. [Drug Delivery Today]
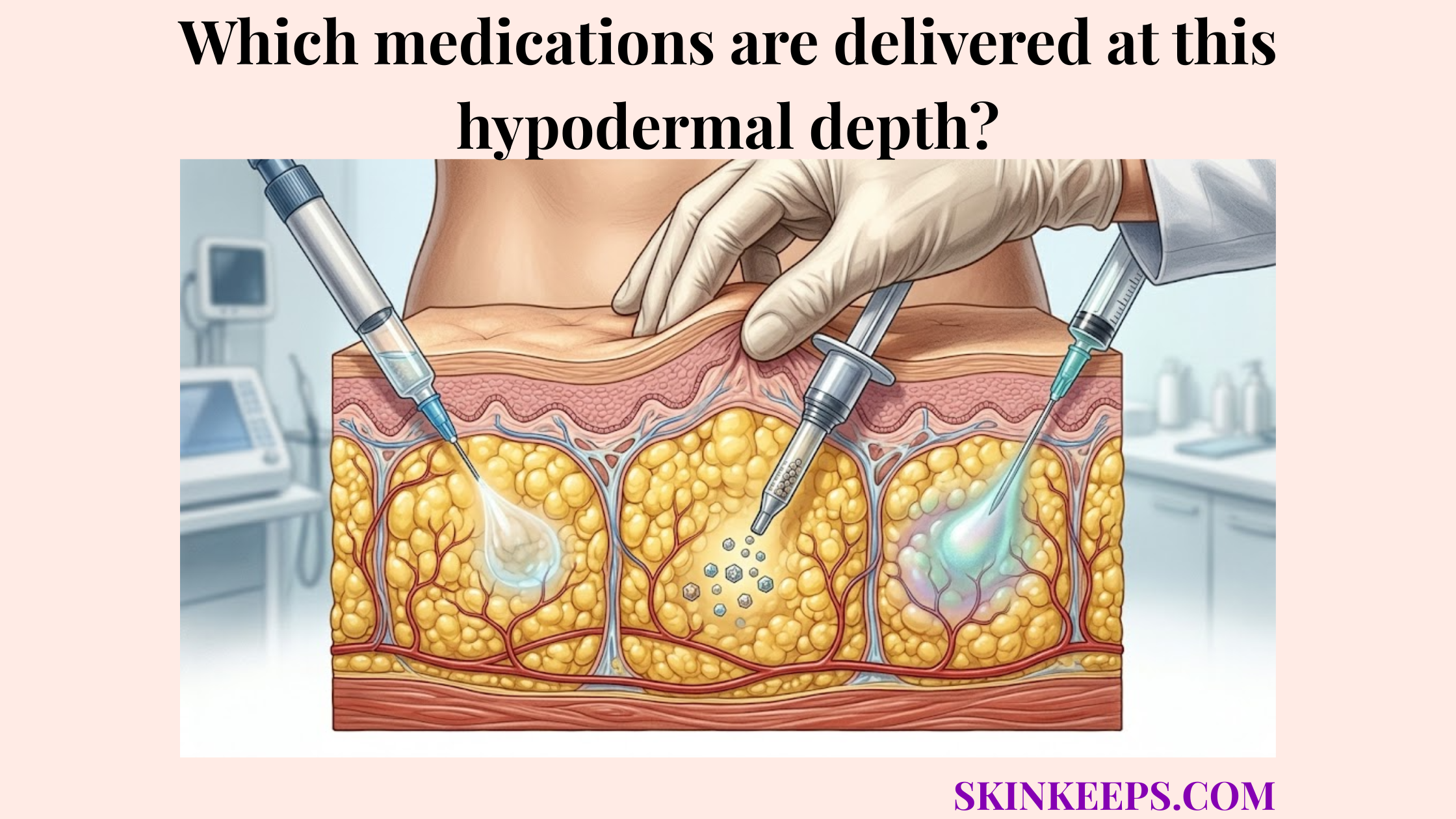
Which pharmaceutical formulations are designed to use deep tissue absorption delays?
Pharmaceutical formulations are designed to use deep tissue absorption delays when the drug product intentionally forms a depot, precipitate, suspension, or slow-dissolving reservoir after subcutaneous administration. These depot formulations leverage the hypodermis to engineer a specific slow-release medication profile.
This formulation design forces the drug to transition slowly from its solid or oily state into the surrounding tissue fluid.
How do insulin glargine microprecipitates delay absorption?
Insulin glargine microprecipitates delay absorption because the acidic solution becomes neutralized in subcutaneous tissue and forms small precipitates that release insulin gradually. This mechanism generates a prolonged basal insulin profile.
EMA product information states that Lantus is soluble at acidic pH 4, and after subcutaneous injection the acidic solution is neutralized, forming microprecipitates from which small amounts are continuously released. [EMA]
How do depot suspensions and oil-based formulations prolong release?
Depot suspensions and oil-based formulations prolong release by slowing drug dissolution or movement from the injection site into systemic circulation. Instead of dissolving instantly, the crystalline suspension or hormone depot must be broken down gradually by local physiological processes.
Consequently, the medication maintains a sustained therapeutic effect over an extended duration of action.
| Drug / Molecule Type | Main Transport Pathway | Absorption Pattern | Safe Writer Language |
|---|---|---|---|
| Small molecules | Interstitial diffusion + capillary uptake | Often faster than larger biologics | Depends on formulation and perfusion |
| Peptides / insulin formulations | Depot behavior + capillary uptake | Formulation-dependent | Absorption varies by insulin type and site |
| Large proteins | Interstitial movement + lymphatic contribution | Often slower and more complex | Lymphatics may contribute more |
| Monoclonal antibodies | Convective transport + lymphatic uptake | Slow systemic appearance | Product-specific pharmacokinetics |
| Depot suspensions | Dissolution-controlled release | Prolonged absorption | Depends on formulation design |
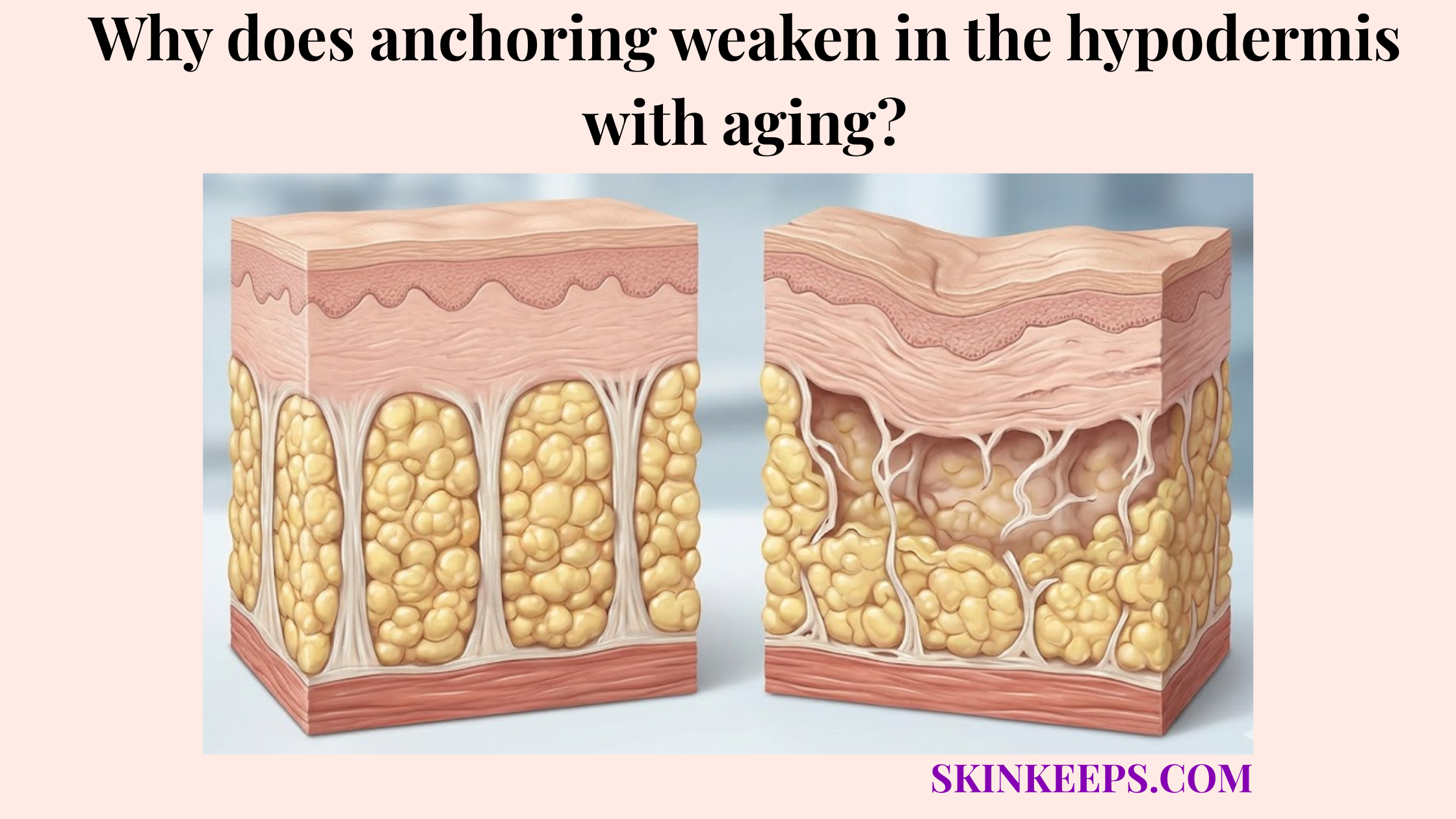
Which pathological conditions unpredictably alter baseline hypodermal pharmacokinetics?
Pathological conditions can unpredictably alter baseline hypodermal pharmacokinetics when they change interstitial fluid volume, local blood flow, tissue pressure, inflammation, or the condition of the injection site. Because hypodermal fat decreases with age and varies with disease, tissue variability directly influences drug uptake.
These unpredictable biological shifts severely compromise expected medication timelines.
How can edema and fluid overload change subcutaneous absorption?
Edema and fluid overload can change subcutaneous absorption by increasing interstitial fluid volume and altering the distance, pressure, and diffusion environment between the drug depot and local vessels. This excess fluid physically widens the gap that molecules must cross.
As a result, the absorption pattern may become significantly delayed or less predictable.
How can severe dehydration or shock reduce absorption?
Severe dehydration or shock can reduce absorption because peripheral vasoconstriction may lower blood flow to skin and subcutaneous tissue. When the body prioritizes internal organs, the hypodermis receives drastically less perfusion.
This lack of blood flow can delay or severely limit the systemic uptake of the injected medication.
How do external temperature and physical exercise accelerate subcutaneous drug dispersion?
External temperature and physical exercise can accelerate subcutaneous drug dispersion when heat, muscle activity, massage, or increased local blood flow changes the movement of medication away from the injection depot. These external actions mechanically or thermally disrupt the stable tissue environment.
For highly sensitive medications like rapid-acting insulin, this disruption can alter the absorption curve entirely.
How does heat-related vasodilation affect absorption?
Heat-related vasodilation can affect absorption because warmer tissue may increase local blood flow and speed uptake for some subcutaneous medications, especially insulin. Heat exposure, such as a hot bath or sauna, expands local capillaries, giving the drug faster access to the bloodstream.
Patients should follow medication-specific guidance to prevent unintended absorption changes.
How do friction, massage, and exercise change absorption?
Friction, massage, and exercise can change absorption by increasing local tissue movement, blood flow, or mechanical dispersion around the injection site. Endotext notes that strenuous exercise of an injected limb within 1 hour can speed insulin absorption, and it also lists vigorous rubbing or local massage as a factor that speeds absorption. [Endotext]
What clinical checklist helps maintain predictable slow-release medication profiles?
A clinical checklist helps maintain predictable slow-release medication profiles by protecting the injection site, avoiding abnormal tissue, following product instructions, and reducing unsupervised behaviors that alter local absorption. Consistent adherence to these protocols prevents unexpected absorption variability.
Practicing safe injection habits ensures the medication performs exactly as intended.
How should environmental and physical factors be managed?
Environmental and physical factors should be managed by avoiding unsupervised heat, cold, rubbing, or strenuous use of the injected area when product instructions or clinician guidance warn that these behaviors can alter absorption. Patients must coordinate exercise timing and temperature exposure with their prescribed treatments.
These simple behavioral adjustments maintain pharmacokinetic stability.
How should injection sites be checked before administration?
Injection sites should be checked before administration because edema, redness, bruising, hardness, infection, scarring, or lipohypertrophy can make absorption less predictable or increase local complication risk. Clinical guidance advises avoiding subcutaneous injection sites that are edematous, bruised, hard, red, broken, infected, over bony areas, or where clothing may rub. [Royal Children’s Hospital Melbourne]
Daily Pharmacokinetic Stability & Absorption Management Checklist
FAQs About Slow Drug Absorption in Subcutaneous Tissue
Why does subcutaneous tissue absorb drugs slowly?
Subcutaneous tissue absorbs many drugs slowly because medication must move through interstitial fluid and extracellular matrix before entering blood or lymphatic vessels. This creates a tissue depot rather than direct bloodstream delivery, but the exact absorption rate depends on drug formulation, site, and local perfusion.
Is slow absorption always guaranteed in the hypodermis?
No. Slow absorption is not guaranteed. Heat, exercise, massage, injection site, lipohypertrophy, tissue swelling, blood flow, drug dose, and formulation can change absorption speed. The route supports slower absorption only when medication and technique match the intended product design.
Do large biologic drugs absorb through lymphatics?
Large biologics often depend more on lymphatic transport than smaller molecules because their size can limit direct blood-capillary uptake. One lymphatic-delivery review reports almost complete lymphatic absorption for proteins around 30–40 kDa in a sheep model. [Drug Delivery Today]
Why does insulin glargine absorb slowly?
Insulin glargine absorbs slowly because its acidic solution becomes neutralized after subcutaneous injection and forms microprecipitates. Small amounts then release gradually, which helps produce a prolonged basal insulin profile under product-specific conditions starting at pH 4. [EMA]
Can heat or massage speed absorption?
Yes, especially for insulin. Endotext lists heat exposure, vigorous massage, and strenuous exercise of the injected area within 1 hour as factors that can speed insulin absorption. Patients should follow medication-specific and clinician-guided instructions. [Endotext]
Can edema make subcutaneous absorption unreliable?
Edema can make subcutaneous absorption less predictable because excess fluid changes tissue pressure, diffusion distance, and the local injection environment. Clinicians often teach patients to avoid swollen, hard, red, bruised, infected, or broken skin sites when injecting.
How can someone keep absorption more predictable?
Absorption is more predictable when the medication is used exactly as prescribed, the correct tissue site is selected, abnormal tissue is avoided, sites are rotated, and post-injection behaviors such as rubbing, heating, or strenuous use follow clinician guidance.
Conclusion
Subcutaneous tissue allows slow drug absorption by creating a tissue depot where medication must move through extracellular matrix, interstitial fluid, capillaries, and lymphatic pathways before reaching systemic circulation. This slow-release behavior is useful only when the medication, formulation, site, and technique are designed for subcutaneous delivery.
At SkinKeeps, we explain skin-layer function through evidence-based anatomy, physiology, and pharmacology. Understanding subcutaneous absorption helps readers respect why product instructions, clinician guidance, site condition, and post-injection behavior matter.