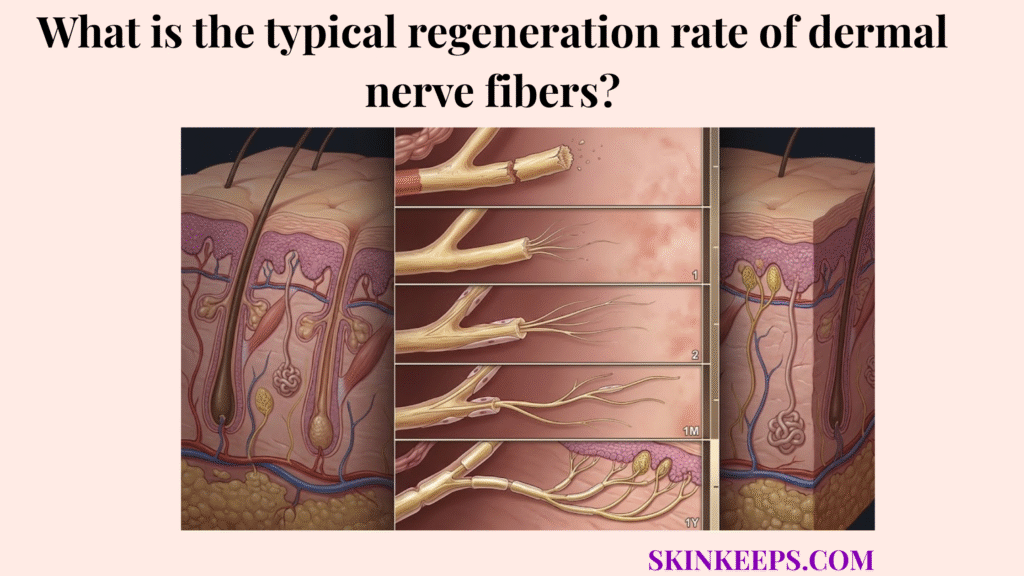
The typical regeneration rate of dermal nerve fibers is about 1 millimeter per day, or roughly 1 inch per month, under favorable conditions, which is why sensory recovery is usually counted in weeks or months rather than days.
This slow pace reflects a biological speed limit dictated by microscopic construction requirements, not just a frustrating healing inconvenience. Closing the skin over a wound happens rapidly through epithelial cell division, but the underlying sensory network must literally rebuild itself from the point of injury down to the fingertips or toes.
Understanding the regeneration rate of dermal nerve fibers requires mapping the biological baseline of 1 mm/day, exploring how injury types disrupt this path, examining the axonal transport limit, assessing metabolic and age-related drag, and identifying supportive clinical protocols.
What is the biological baseline for the regeneration rate of dermal nerve fibers?
The biological baseline for the regeneration rate of dermal nerve fibers is that peripheral axons regrow slowly, commonly around 1 mm per day, because new nerve material must be built and advanced progressively from the surviving neuron toward the target tissue.
Peripheral nerve repair is a construction project. A severed axon must push an exploratory structure—the growth cone—through the tissue bed. This advancing tip senses chemical cues and lays down new biological track millimeter by millimeter.
Because the process requires physical extension rather than simply sealing two edges together, clinical recovery is dictated by the total distance the axon must cover to achieve reinnervation (reconnecting with the sensory target in the skin).
Multiple peripheral-nerve reviews by Baradaran et al. (2021) and Grinsell and Keating (2014) describe a practical clinical recovery rate of about 1 mm per day or 1 inch per month in humans. A useful milestone map is:
- 24 hours → about 1 mm
- 1 week → about 7 mm
- 1 month → about 30 mm
- 1 year → about 365 mm
These are expectation-setting estimates, not guarantees for every case [PMC] [PMC].
How do injury types influence the regeneration rate of dermal nerve fibers?
Injury types influence the regeneration rate of dermal nerve fibers because injuries that preserve internal guidance architecture usually allow cleaner regrowth than injuries that fully disrupt the pathway.
In a crush injury, the axon inside the nerve is damaged, but the outer protective endoneurial tube remains intact. This preserved pathway acts as a perfectly aligned tunnel, guiding the regenerating axon straight back to its original target in the dermis.
In a transection injury (where the nerve is completely severed), the entire structure is cut. The regrowing axon must now navigate a physical gap, battle scar tissue, and rely on newly formed Schwann-cell guidance pathways. This severely stalls the overall recovery pace, even if the daily biological growth rate is identical, often leading to misrouted fibers.
How does axonal transport limit the regeneration rate of dermal nerve fibers?
Axonal transport limits the regeneration rate of dermal nerve fibers because a regenerating axon cannot advance faster than it can receive the proteins, membranes, organelles, and signaling machinery needed at its growth cone.
The nerve cell body (located near the spinal cord) acts as the manufacturing plant. It must assemble the raw materials required for growth and ship them down the length of the axon to the advancing tip. This physical logistics network represents a hard biological bottleneck.
No matter how optimal the external environment is, the growth cone cannot surge forward if the slow axonal transport system cannot deliver structural proteins fast enough to build the extending membrane.
How does metabolic health dictate the regeneration rate of dermal nerve fibers?
Metabolic health dictates the regeneration rate of dermal nerve fibers because successful regrowth depends on blood flow, oxygen delivery, nutrient support, and a microenvironment that does not chronically damage the nerve.
Regeneration requires massive amounts of ATP (cellular energy). If microvascular support is compromised, the regenerating tip starves, causing recovery to stall entirely. Good circulation is non-negotiable for sustained daily growth.
This is why recognizing which factors slow dermal nerve recovery helps explain why conditions like diabetic neuropathy severely slow down the process. Elevated blood sugar damages the fragile micro-vessels feeding the nerve, stripping the growth cone of its essential oxygen and nutrient supply and often causing the regeneration process to fail.
How do life stages compare regarding the regeneration rate of dermal nerve fibers?
Life stages compare differently because the regeneration rate of dermal nerve fibers tends to be more efficient in younger tissue and more impaired in older tissue with reduced repair capacity.
Age-related repair decline affects the speed of macrophage clearance, the responsiveness of Schwann cells, and overall metabolic vigor. While a young child may recover rapidly and completely from a nerve injury, an older adult faces a much slower biological timeline and a higher risk of incomplete sensory return.
Older tissue simply mobilizes the necessary repair machinery less robustly, stretching the 1 mm/day expectation significantly.
| Age group | Average regeneration tendency | Functional recovery pace | Main limiting factor |
|---|---|---|---|
| Children / younger adults | Faster and more efficient | Better overall recovery | Lower chronic biological drag |
| Middle age | Moderate | Variable | Rising metabolic and vascular limits |
| Older adults | Slower and less efficient | Delayed or incomplete recovery more common | Reduced circulation and slower repair biology |
How does recovery distance change the regeneration rate of dermal nerve fibers in practice?
Recovery distance changes the practical timeline of the regeneration rate of dermal nerve fibers because even when daily advance is similar, longer distances turn the same biological pace into much longer overall recovery.
If a nerve is injured at the wrist, it only needs to travel a short distance to reinnervate the fingertip. If the same nerve is injured near the shoulder, it must cover a massive anatomical gap at the same 1 mm/day speed limit.
Because the daily speed is relatively fixed, a proximal injury guarantees a months-long (or years-long) delay before sensory return occurs at the dermal surface. This practical reality highlights why assessing the injury site is crucial when asking can full sensory function return after dermal nerve damage?
What clinical protocols optimize the regeneration rate of dermal nerve fibers?
Clinical protocols optimize the regeneration rate of dermal nerve fibers by improving the growth environment, preserving circulation, supporting metabolism, and using structured therapies that may enhance regenerative signaling without bypassing biology.
Optimization centers on removing the barriers that slow growth—such as inflammation or metabolic starvation—rather than attempting to force the nerve to grow at an impossible speed.
How do B-complex vitamins and Alpha-Lipoic Acid support the regeneration rate of dermal nerve fibers?
B-complex vitamins and Alpha-Lipoic Acid may support the regeneration rate of dermal nerve fibers by improving the metabolic and oxidative environment needed for peripheral nerve maintenance and regrowth, but they should be framed as supportive measures rather than instant accelerators.
Vitamin B12 acts as a vital co-factor in myelin synthesis and neuronal energy production, while B6 provides secondary metabolic support. Alpha-lipoic acid helps neutralize oxidative stress in the damaged tissue, which is well-documented for supporting symptom management in neuropathic pain, creating a more favorable environment for the nerve to execute its slow recovery without inflammatory interference.
How does electrical stimulation support the regeneration rate of dermal nerve fibers?
Electrical stimulation may support the regeneration rate of dermal nerve fibers by enhancing the regenerative environment and, in some settings, improving axon growth and sensorimotor recovery.
Structured, low-frequency electrical stimulation applied shortly after injury has been shown clinically to upregulate crucial neurotrophic factors. It acts as a targeted physical therapy signal for the nerve, promoting directional growth and potentially reducing the time axons stall before crossing the injury gap.
Optimization Framework
- Problem → nerve regrowth is biologically slow and vulnerable to stalling.
- Implication → sensory recovery may take months or become incomplete.
- Solution → improve metabolic support, preserve circulation, and use structured therapies where appropriate.
What are the summary facts for the regeneration rate of dermal nerve fibers?
The summary facts are that the regeneration rate of dermal nerve fibers is usually slow, commonly approximated at about 1 mm/day, and strongly shaped by injury pattern, distance, age, and metabolic support.
Regeneration Facts Checklist
What steps can you take to support the regeneration rate of dermal nerve fibers today?
The regeneration rate of dermal nerve fibers is best supported when patients protect the repair pathway, optimize metabolic health, and judge recovery by sensory return rather than skin appearance alone.
Recovery Support Execution Checklist
Quick Answers About the Regeneration Rate of Dermal Nerve Fibers
What is the usual regeneration rate of dermal nerve fibers?
The typical regeneration rate of dermal nerve fibers is approximately 1 millimeter per day, or about 1 inch per month, under healthy biological conditions. Althagafi and Nadi (2023) confirm this rough clinical rule for peripheral axonal growth [StatPearls].
Why is nerve recovery measured in millimeters per day?
Nerve recovery is measured in millimeters per day because regenerating axons must physically build new structural material and push it forward microscopically from the injury site to the target tissue.
Do crush injuries recover faster than severed injuries?
Yes. Crush injuries generally recover faster and more completely because the internal endoneurial tubes remain intact, providing a clear, uninterrupted guidance pathway for the regrowing axon.
Why does skin heal before sensation returns?
Skin heals before sensation returns because epidermal cells divide and close a wound in weeks, whereas a severed nerve must regrow millimeter by millimeter over distances that often take months.
Does diabetes slow nerve regrowth?
Yes. Diabetic neuropathy and poor glycemic control impair microvascular circulation, reducing the vital oxygen and metabolic support required to sustain the energy-intensive process of axonal regrowth.
Does age reduce nerve repair efficiency?
Yes. Aging diminishes Schwann-cell responsiveness and slows metabolic repair biology, making nerve regeneration less efficient and functional sensory return less complete in older adults.
Can electrical stimulation improve nerve recovery?
Clinical electrical stimulation applied early after injury may enhance the regenerative environment by upregulating neurotrophic factors, thereby supporting more efficient axonal growth.
Do B vitamins or alpha-lipoic acid speed nerve repair?
B-complex vitamins and alpha-lipoic acid support the metabolic and oxidative environment needed for nerve maintenance, but they act as supportive nutritional measures rather than magical accelerators that bypass biological speed limits.
Conclusion
In conclusion, the regeneration rate of dermal nerve fibers is slow because peripheral nerve repair is a supply-limited biological construction process, not a simple wound-closing event.
By understanding the 1 mm/day rule, acknowledging the bottleneck of axonal transport, and recognizing that injury architecture and distance heavily dictate the recovery timeline, we can align our expectations with biological reality. While we cannot erase this natural speed limit, employing targeted metabolic support and protective therapies ensures the optimal environment for the nerve to successfully complete its long journey back to the skin.