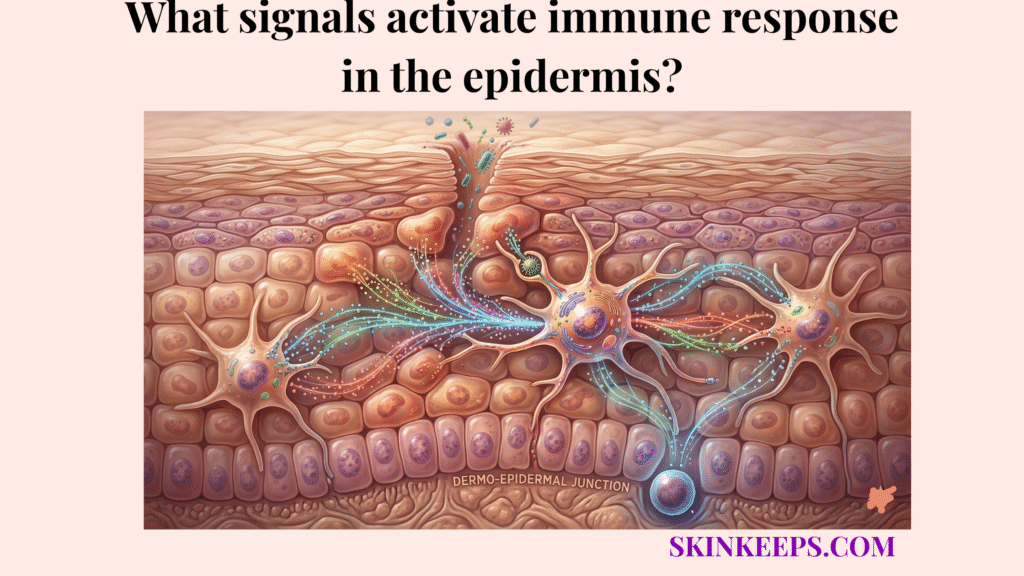
The signals that activate immune response in the epidermis include microbial danger signals, damage-associated stress signals, and locally released inflammatory mediators such as alarmins, cytokines, and chemokines. These signals alert epidermal cells that the barrier has encountered a pathogen, injury, or immune disturbance and needs to mount a defense.
The epidermis acts early by sensing foreign patterns and tissue stress, translating these cues into a coordinated local response long before an infection becomes systemic. This proactive signaling ensures that the skin remains an active surveillance organ rather than a passive boundary.
Understanding what signals activate immune response in the epidermis makes it easier to explain why the skin can react quickly to microbes and injury, how local immune activation begins, and what happens when these signals become too weak or too strong.
Why do activating signals matter in epidermal immune defense?
Activating signals matter in epidermal immune defense because they allow the skin to convert localized environmental exposure into a rapid defensive response before pathogens can penetrate deeper tissue.
Immune activation depends on fast signal interpretation rather than waiting for large-scale tissue destruction. By recognizing molecular “red flags” immediately, the epidermis can recruit localized reinforcements to neutralize threats without needing to trigger a full-body fever or systemic response.
Epidermal signaling matters because the skin must react at the point of contact, using molecular cues rather than delayed structural failure. This speed is a biological necessity; the skin hosts about one million bacteria per square centimeter, making constant low-level surveillance the baseline for health (PMC).
What microbial signals activate immune response in the epidermis?
Microbial signals that activate immune response in the epidermis consist of pathogen-associated molecular patterns (PAMPs)—conserved microbial signatures recognized as foreign—such as bacterial cell-wall components and viral nucleic-acid patterns that the skin identifies as foreign.
These microbial cues are useful because they let the epidermis detect foreign organisms without needing to identify every species individually. By focusing on “shared” molecular traits of pathogens, the skin can maintain a broad and efficient detection net.
Bacterial, viral, and fungal organisms expose different pattern classes, but all function as non-self triggers for epidermal recognition systems. This ensures that the defense system is responsive to a wide variety of biological invaders (ScienceDirect).
What damage signals activate immune response in the epidermis?
Damage signals activate immune response in the epidermis when stressed or injured cells release damage-associated molecular patterns (DAMPs)—self-derived danger signals released by stressed tissue—alerting the skin to barrier disruption even when live pathogens are not the main trigger.
The epidermis treats mechanical stress, UV injury, and barrier disruption as urgent immune-relevant events. This “sterile inflammation” allows the maqaarka to begin repair and protection protocols even in the absence of an infection.
Extracellular ATP and HMGB1 are canonical examples of DAMP-like signals in skin biology. Keratinocytes actively release ATP in response to brief external stress, while HMGB1 is discharged after UV-associated injury to trigger inflammatory readiness (PMC).
| Condition | Primary DAMP Signal | Consequence |
|---|---|---|
| Barrier disruption | Extracellular ATP | Immediate stress alert |
| UV-induced injury | HMGB1 release | Inflammatory readiness |
| Cellular stress | Intracellular molecules | Local signaling activation |
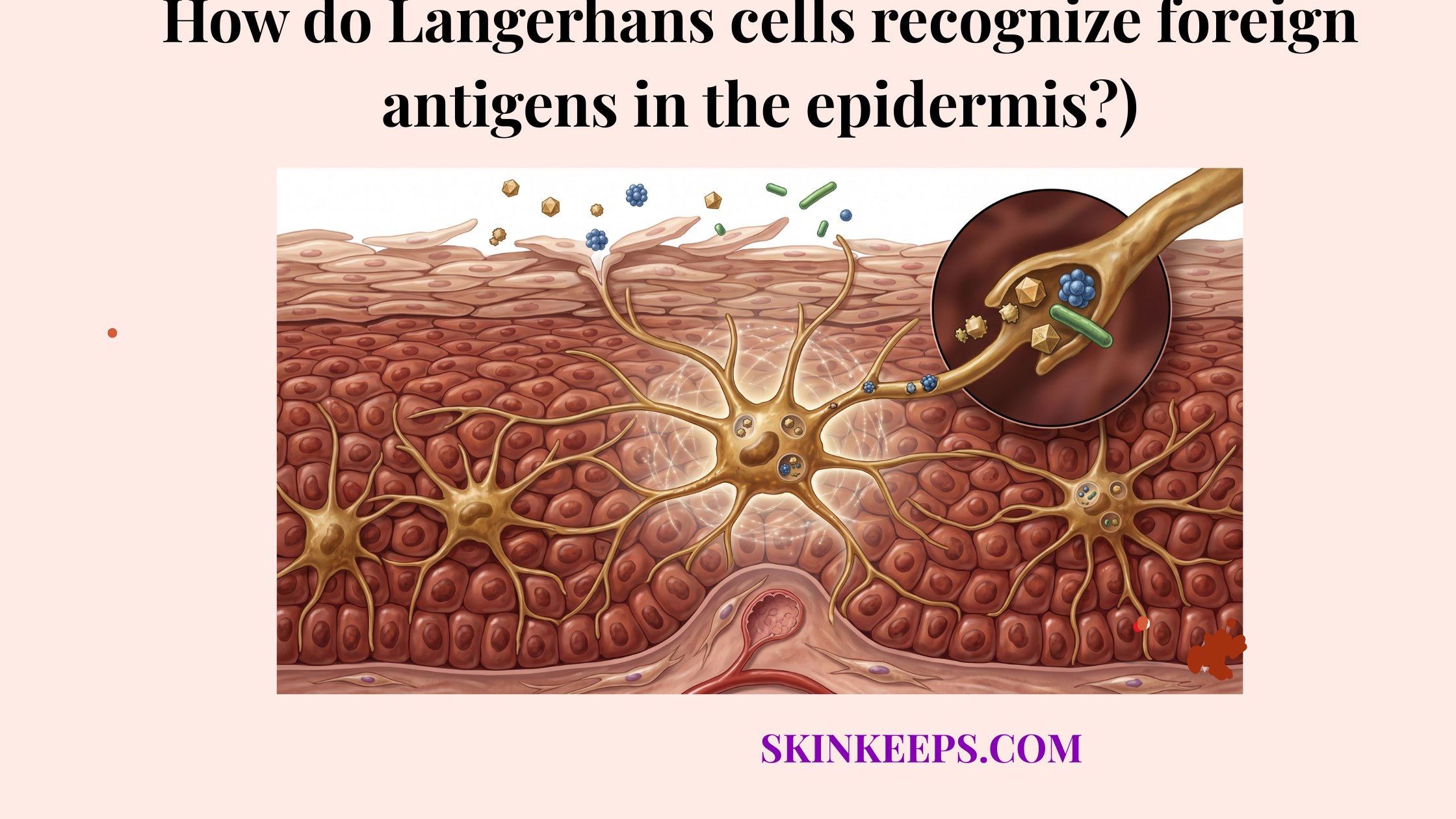
How do recognition systems convert microbial and damage signals into epidermal immune activation?
Recognition systems convert microbial and damage signals into epidermal immune activation by using Pattern Recognition Receptors (PRRs)—immune-sensing molecules—and Toll-like receptors (TLRs)—a major PRR family—to bind danger patterns and trigger intracellular cascades.
Receptor engagement is the critical step that converts environmental exposure into active biology. The most vital switch in this cascade is NF-κB, a major transcriptional switch that links receptor recognition to the expression of inflammatory genes inside the cell nucleus.
Primary keratinocyte studies show functional TLR-linked responses to PAMPs, resulting in the enhanced release of mediators such as TNF-α and CXCL8 after receptor stimulation. This converts a surface encounter into a deep cellular alarm (ScienceDirect).
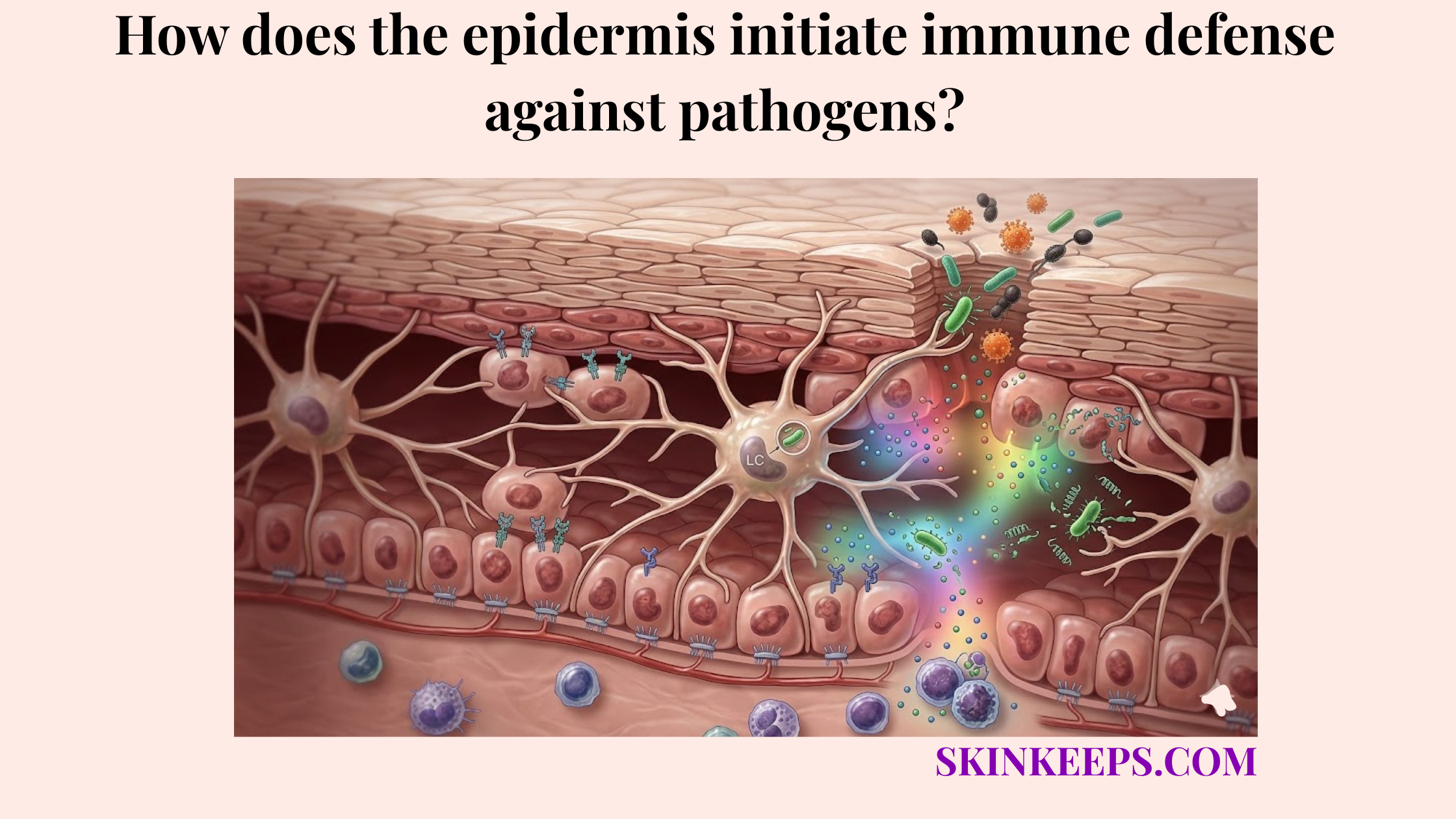
How do alarmins activate immune response in the epidermis?
Alarmins—rapidly released distress mediators that alert neighboring cells—activate immune response in the epidermis by acting as rapid distress molecules released during cell injury.
Alarmins matter because they make local injury visible to neighboring cells long before slower genetic programs can synthesize new cytokines. They are essentially “emergency flares” that are pre-packed and ready for immediate deployment.
IL-1α is especially important as an early alarmin, while IL-33 is strongly linked to stress- and wound-associated signaling. Keratinocytes actively release IL-1α after membrane disruption to maintain danger signaling in the skin (Nature).
How do cytokines and chemokines amplify immune response in the epidermis?
Cytokines and chemokines amplify immune response in the epidermis by broadcasting instructions that intensify the local defensive state and direct the movement of recruited immune cells.
In this amplification sequence, Cytokines act as inflammatory messenger proteins that shape the magnitude of the response, while Chemokines function as directional signaling molecules that guide immune-cell movement toward the injury site.
How do cytokines activate and shape epidermal immune response?
Cytokines regulate the intensity and character of the inflammatory state after the first recognition event. Primary keratinocyte studies show that inflammatory stimulation drives the production of mediators like IL-6 and TNF-α through NF-κB-linked programs, which is central to sustained amplification (Nature, 2020).
How do chemokines direct immune-cell movement after epidermal activation?
Chemokines direct immune-cell movement by establishing concentration gradients that pull specific immune populations toward the inflamed site. Keratinocyte-derived CCL20 is a major recruitment chemokine, while CXCL8 is strongly linked to neutrophil recruitment in keratinocyte-driven settings (Nature, 2020).
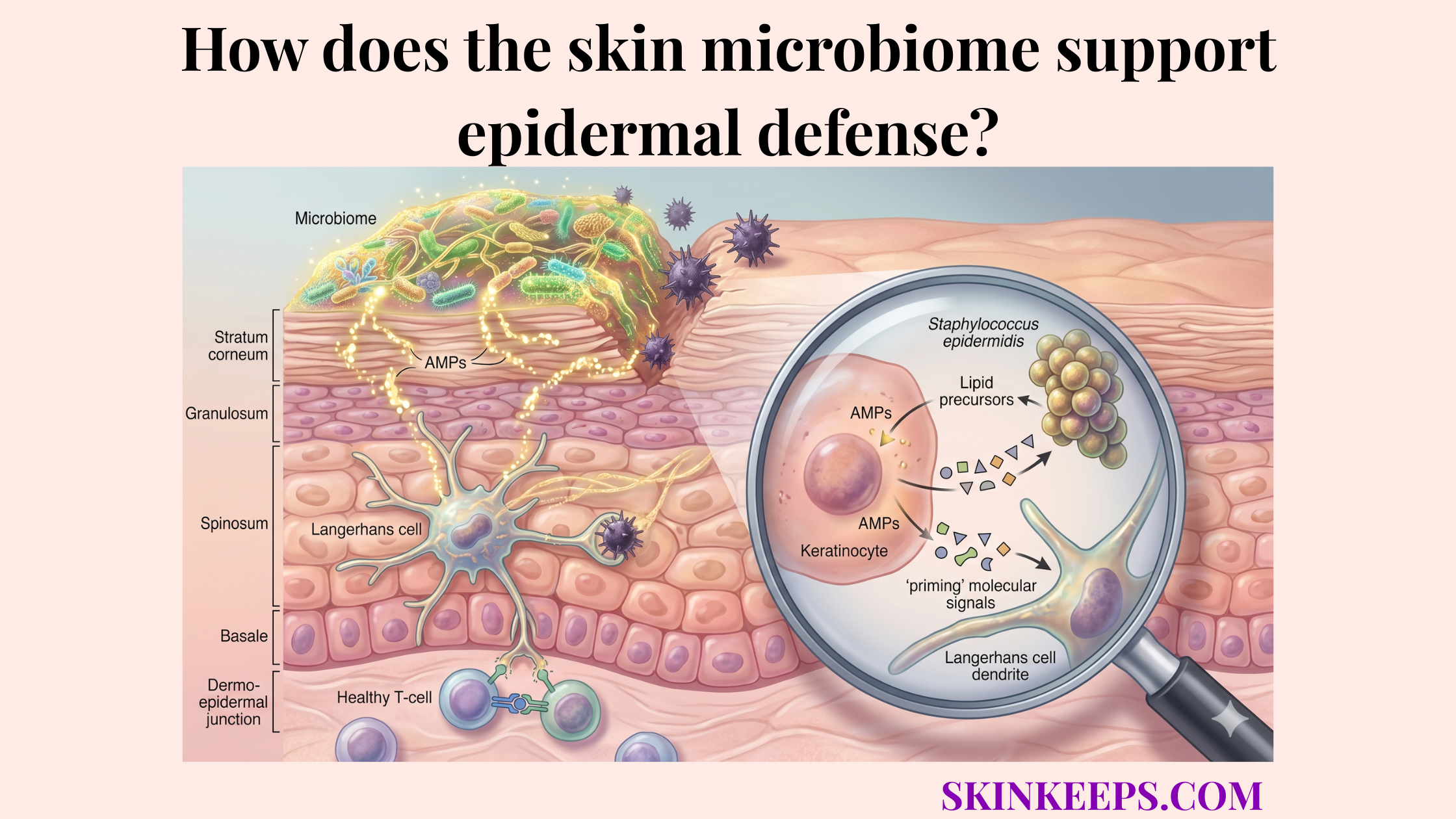
How do antimicrobial peptides fit into the signals that activate epidermal immune response?
Antimicrobial peptides (AMPs)—small proteins that act as direct chemical effectors—fit into the signals that activate epidermal immune response as downstream molecules that are induced after danger signals switch keratinocytes into defensive mode.
AMPs are not the initial signal themselves; they are the “weaponry” deployed by the cells after the alarm has been heard. Keratinocyte injury and inflammatory activation rapidly increase AMP-associated programs, linking danger recognition to direct action against pathogens (PMC).
How does protective epidermal immune signaling differ from chronic inflammatory signaling?
Protective epidermal immune signaling differs from chronic inflammatory signaling because protective signaling resolves after the threat is contained, whereas chronic signaling persists after the danger has been neutralized.
| Signal Pattern | Trigger Style | Immune Effect | Skin Outcome |
|---|---|---|---|
| Protective | Short-lived | Controlled activation | Defense & recovery |
| Chronic | Persistent | Ongoing amplification | Redness & sensitivity |
How can you tell when epidermal immune-activating signals are dysregulated?
You can tell when epidermal immune-activating signals are dysregulated by the presence of chronic unexplained redness, frequent stinging with basic products, and recurring barrier instability.
Warning Signs of Dysregulated Signaling
What factors can weaken or overactivate epidermal immune-activating signals?
Healthy epidermal immune-activating signals are most commonly disrupted by chronic barrier depletion, excessive UV radiation, and repetitive chemical or mechanical irritation that keep inflammatory pathways abnormally engaged.
UV exposure is especially disruptive because it can alter both signaling molecules and epidermal immune-cell behavior. In human UVB studies, the depletion of Langerhans cells after irradiation is linked mainly to forced migration, showing how UV can remodel signaling architecture (ScienceDirect).
What habits help support balanced immune signaling in the epidermis?
Habits help support balanced immune signaling in the epidermis by prioritizing barrier integrity and environmental protection to reduce the background noise of unnecessary damage signals.
How does barrier support help maintain balanced epidermal immune signaling?
Barrier support reduces the repeated tissue stress that drives false-alarm damage signaling. Because keratinocytes release extracellular ATP in response to barrier perturbation, support is a way to reduce repeated danger signaling rather than just a comfort step (PLOS ONE).
How does sun protection help preserve healthier epidermal immune signaling?
Sun protection helps preserve healthier epidermal immune signaling by reducing UV-driven damage cues and downstream immune distortion. Primary studies show that UVB exposure reshapes cutaneous signaling by altering cell populations and cytokine thresholds (ScienceDirect).
How do gentler routines reduce unnecessary epidermal immune activation?
Gentler routines reduce unnecessary epidermal immune activation by eliminating repetitive mechanical stress that keeps inflammatory pathways triggered. Avoiding over-cleansing helps prevent a self-sustaining loop of barrier stress and signal noise (PLOS ONE).
What are the key takeaways about the signals that activate immune response in the epidermis?
The key takeaways center on the rapid conversion of environmental cues into molecular alarms and the necessity of proper shutdown after defense.
- ● Immune response is activated by PAMPs, DAMPs, and local mediators.
- ● PRRs and TLRs convert these patterns into alarmin and cytokine cascades.
- ● AMPs provide direct chemical defense once signaling begins.
- ● Healthy skin depends on signals that activate quickly and resolve completely.
What daily steps can you take to support healthy immune activation in the epidermis?
You can support healthy immune activation in the epidermis daily by reducing routine-based irritation and shielding the skin from cumulative UV and barrier damage.
Daily Immune Signaling Protocol
Healthy epidermal immunity depends on the right signals activating at the right time—not on the skin living in a permanent state of alarm.
Build your routine around barrier stability and controlled immune balance if your goal is stronger, calmer, more resilient skin defense.
Ultimately, when you reduce signal noise, you improve the precision of your skin’s defense.