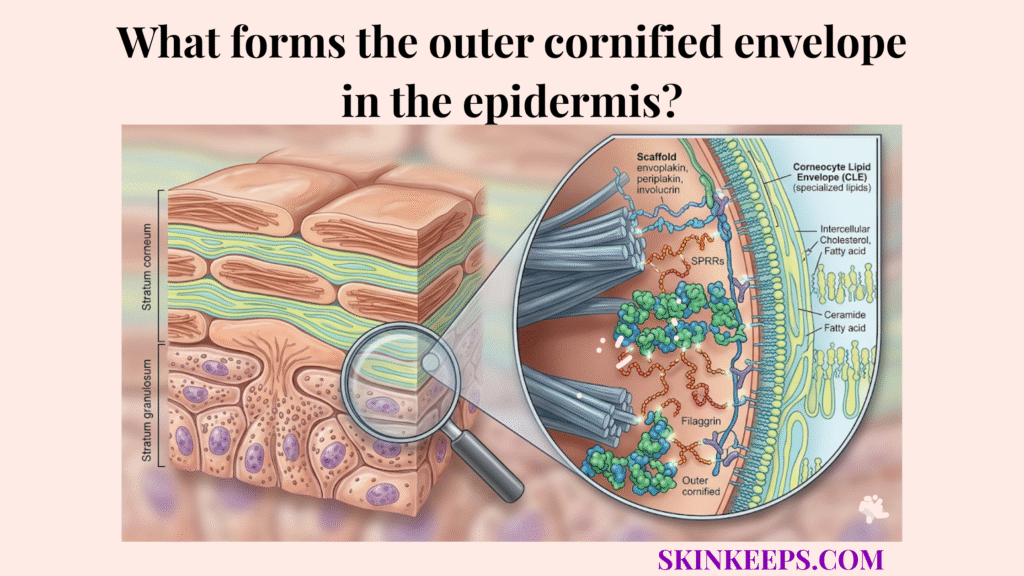
The outer cornified envelope in the epidermis is formed by the staged assembly of a cross-linked protein scaffold and its final lipid reinforcement, creating the highly resistant shell that replaces the keratinocyte plasma membrane during terminal differentiation. This outer shell is built through a sequence of scaffold placement, enzymatic cross-linking, bulk protein reinforcement, and final lipid attachment.
The cornified envelope is the decisive structural transition that turns a living cell boundary into an insoluble, chemically resistant barrier surface. This specialized armor ensures that as cells arrive at the surface, they are biologically equipped to withstand physical and chemical stress.
Understanding what forms the outer cornified envelope in the epidermis makes it easier to explain how the skin builds its final protective shell, why defective assembly weakens barrier performance, and what structural steps are required for successful envelope formation.
Why is cornified envelope formation essential for epidermal permeability?
Cornified envelope formation is essential for epidermal permeability because it transforms the outermost living-cell boundary into a durable, chemically resistant barrier structure that is far more resilient than an ordinary plasma membrane.
Surface cells must resist water loss, chemical penetration, and mechanical stress at the same time, which is why the membrane has to be replaced by a tougher shell. This compaction is the primary mechanism that prevents barrier disruption from increasing TEWL, the invisible evaporation of moisture through the skin surface.
The cornified envelope is commonly described as an approximately 15-nm-thick insoluble structure, and classic biochemical studies showed that envelope material remains resistant even after boiling in sodium dodecyl sulfate-containing solutions (PMC).
| Component | Nature | Functional Result |
|---|---|---|
| Plasma Membrane | Lipid bilayer | Too fragile for surface duty |
| Cornified Envelope | Insoluble protein shell | Lower permeability and high resistance |
How does cornified envelope formation begin with involucrin and periplakin?
Cornified envelope formation begins with involucrin and periplakin aligning along the inner surface of the plasma membrane to establish the first structural template for later protein reinforcement and cross-linking.
In this early phase, involucrin acts as the initial scaffolding protein that provides the essential template, while periplakin and envoplakin serve as membrane-proximal markers that help align the scaffold precisely at the cell edge.
The earliest envelope phase is about localization: certain proteins must first mark the future periphery before later reinforcement can occur. Understanding why keratinocytes lose their nuclei during this stage explains how the cell sacrifices its living functions to focus on building this peripheral scaffold.
Periplakin and envoplakin are recognized CE precursors that localize to the plasma membrane and desmosomal regions during assembly, ensuring the shell is anchored where it is needed most (PNAS).
How do transglutaminases catalyze cornified envelope formation?
Transglutaminases catalyze cornified envelope formation by creating covalent Nε-(γ-glutamyl)lysine isopeptide bonds that transform a loose protein scaffold into a mechanically stable, insoluble barrier shell.
In this chemical transition, transglutaminases function as specialized enzymes that catalyze the cross-linking of proteins. This process relies on isopeptide bonds, which are incredibly strong covalent chemical bonds that provide the extreme durability required for the final armor.
Transglutaminase-driven cross-linking is the event that changes a collection of keratin filaments into a true shell. Epidermal transglutaminases are calcium-dependent enzymes, and TG1, TG3, and TG5 are all implicated in cornified envelope construction (MDPI).
How does TG1 contribute to cornified envelope formation?
TG1, or Transglutaminase 1, contributes to cornified envelope formation by facilitating the membrane-proximal cross-linking of early proteins, effectively anchoring the initial scaffold at the cellular periphery. It serves as the primary enzyme that converts a loose template into an insoluble fixed platform.
How do TG3 and TG5 strengthen cornified envelope formation?
TG3 and TG5 strengthen cornified envelope formation by contributing additional cross-linking during later maturation stages, reinforcing the growing protein network for greater durability. These enzymes densify the already-forming structure into a solid matrix suitable for barrier protection (MDPI).
How does loricrin complete the protein scaffold during cornified envelope formation?
Loricrin completes the protein scaffold during cornified envelope formation by supplying the major bulk of the mature shell, creating the final thickness and structural resilience required for barrier survival.
In this reinforcement phase, loricrin acts as the major protein component that provides the mature envelope’s bulk, while Small Proline-Rich Proteins (SPRRs) serve as smaller reinforcing units that fine-tune the shell’s density and flexibility.
Involucrin and plakin proteins help start the shell, but loricrin provides most of the mature mass. Loricrin is widely described as the major protein component of the mature cornified envelope, contributing more than 70% of its protein mass in normal epidermis (PMC).
How does lipid linkage make cornified envelope formation hydrophobic?
Lipid linkage makes cornified envelope formation hydrophobic by covalently attaching ω-hydroxyceramides to the mature protein scaffold, creating the corneocyte lipid envelope (CLE) that helps restrict water movement.
In this waterproofing step, ω-hydroxyceramides are specialized lipids that form the permanent bond needed to make the shell water-repellent. The resulting CLE is the critical lipid layer bound to the protein shell that ensures the overall barrier remains impermeable.
A protein shell alone is not enough; the outer structure also needs hydrophobic reinforcement to effectively repel water and block irritants. ω-hydroxyceramides are required for CLE formation and normal epidermal permeability-barrier function (ScienceDirect).
How do failures in cornified envelope formation cause barrier defects?
Failures in cornified envelope formation cause barrier defects because the epidermis depends on a flawless sequence of scaffolding, cross-linking, and lipid integration to maintain moisture control.
A primary clinical example is TGM1 deficiency, a genetic absence or malfunction of the TG1 enzyme that is a well-established cause of autosomal recessive congenital ichthyosis. This failure produces absent or defective cornified envelopes with impaired barrier function (PMC).
| Failed Stage | Structural Consequence | Clinical Direction |
|---|---|---|
| Scaffold failure | Poor peripheral template | Incomplete shell assembly |
| Cross-linking failure | Fragile insoluble shell | TGM1-linked ichthyotic defects |
| Lipid-linkage failure | Poor hydrophobic reinforcement | Chronic dryness and flaking |
What are the key milestones in successful cornified envelope formation?
Successful cornified envelope formation requires accurate scaffold alignment, effective transglutaminase-driven isopeptide cross-linking, and final lipid attachment so the epidermis can produce a chemically resistant barrier shell.
Final CE Validation Checklist
Use this framework to analyze epidermal barrier assembly as a staged construction process rather than a loose list of proteins and enzymes.
Ultimately, when you understand the assembly of the cornified envelope, you understand the foundational architecture of human skin.