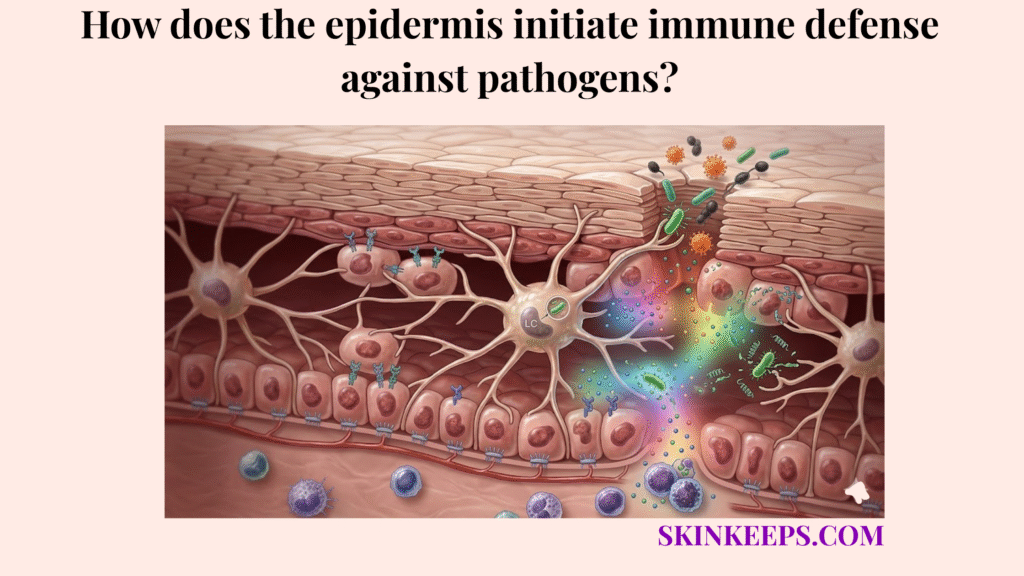
The epidermis initiates immune defense against pathogens by detecting microbial danger signals through pattern recognition receptors, then activating keratinocyte signaling, antimicrobial peptide release, and immune-cell communication to halt infection at the site of contact. This process allows the skin to respond with extreme speed before a localized breach spreads to deeper tissues.
The epidermis is an active immune interface, not just a physical barrier, constantly monitoring for bacteria, viruses, and fungi through resident epithelial and immune signaling systems. Keratinocytes and resident immune cells are now widely recognized as active participants in cutaneous innate defense.
Understanding epidermal immune defense initiation makes it easier to explain how the skin detects pathogens, how keratinocytes launch the first response, how resident immune cells coordinate escalation, and what goes wrong when the system becomes chronically inflamed or immunologically weak.
Why is epidermal immune defense initiation critical for cutaneous homeostasis?
Epidermal immune defense initiation is critical for cutaneous homeostasis because the skin must function as an active surveillance organ that identifies and neutralizes microbial threats before they can trigger deeper inflammatory cascades.
A fast local response is necessary because the skin surface is continuously exposed to microbes and environmental injury, yet still has to preserve tissue stability. This intelligence-led defense prevents every minor scratch from becoming a systemic infection.
The skin surface is commonly described as hosting about one million bacteria per square centimeter, which is why constant low-level immune surveillance is biologically necessary. Understanding how the skin microbiome supports epidermal defense—the diverse community of microorganisms living on the skin surface—clarifies how beneficial bacteria help regulate this surveillance baseline (PMC).
| Environmental Reality | Defense Requirement | Outcome |
|---|---|---|
| Constant microbial contact | Active PRR surveillance | Infection prevention |
| Surface breaches | Fast local detection | Containment of threats |
| Tissue stability | Controlled early response | Preserved homeostasis |
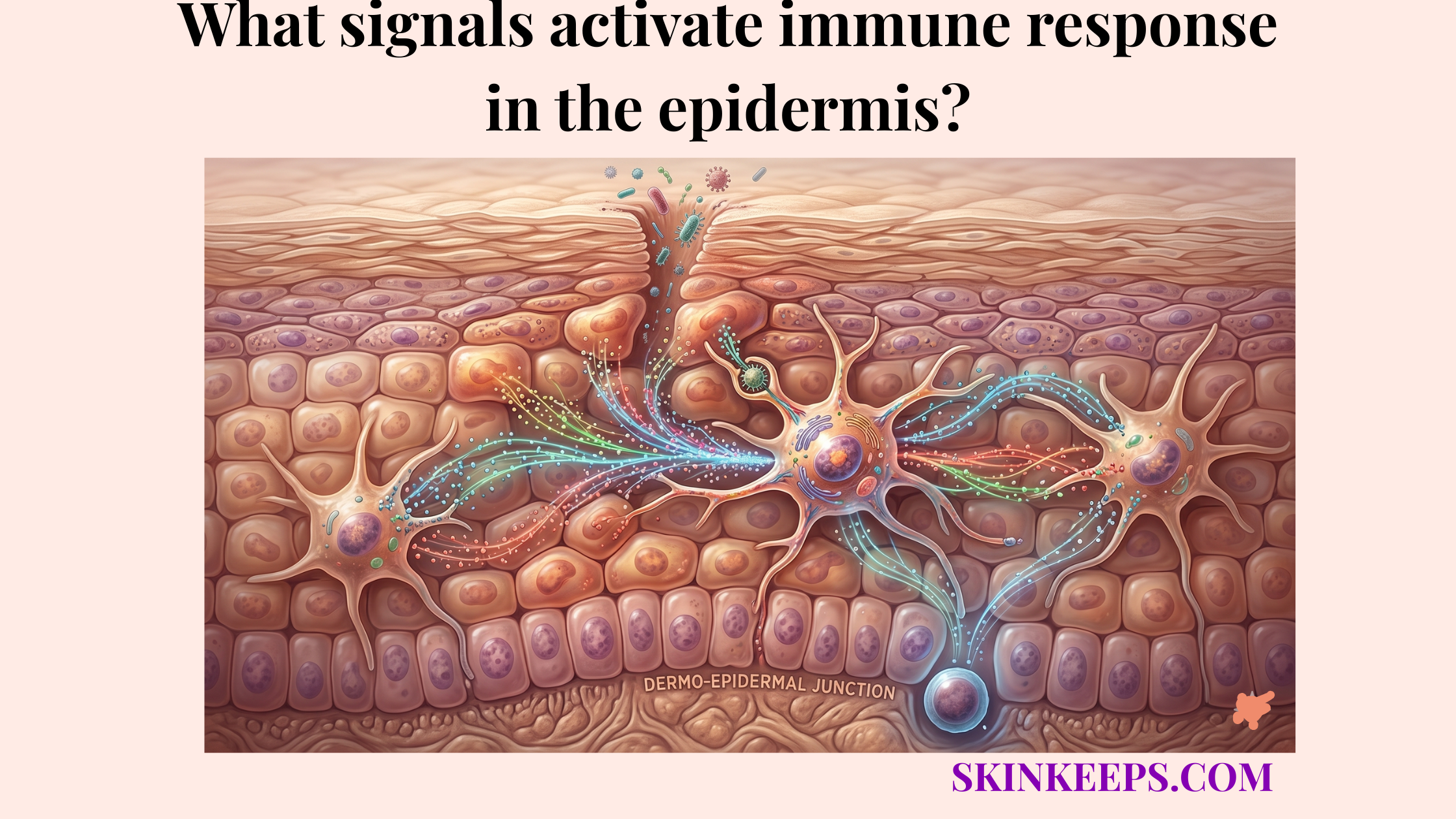
How does epidermal immune defense initiation begin through pattern recognition receptors?
Epidermal immune defense initiation begins through pattern recognition receptors (PRRs)—proteins used by host cells to identify microbial molecules—especially Toll-like receptors, which detect conserved microbial components and convert surface contact into intracellular alarm signaling.
PRR biology matters because the epidermis does not wait for obvious infection; it reacts to Pathogen-Associated Molecular Patterns (PAMPs)—conserved molecular signatures shared by pathogens—as soon as they appear. The speed of the startup response depends heavily on what signals activate immune response in the epidermis during this initial molecular recognition phase.
Human keratinocytes are widely reported to express multiple Toll-like receptors (TLRs)—a major class of PRRs that detect specific microbial components—most consistently TLR1–6, TLR9, and TLR10, which gives the epidermis broad sensing capacity for bacterial, viral, and fungal signatures (PMC).
How do keratinocytes trigger epidermal immune defense initiation?
Keratinocytes—the primary epithelial cells of the epidermis—trigger epidermal immune defense initiation by acting as frontline sentinels that detect damage or microbial contact and then release alarmins, cytokines, and antimicrobial mediators that alert the broader cutaneous immune system.
Keratinocytes matter not only because they form the barrier, but because they determine how fast and how intensely the early immune response is broadcast. They are the “broadcast towers” of the skin’s defense.
How do keratinocytes function as frontline immune sentinels?
Keratinocytes function as frontline immune sentinels by using their position in the upper epidermis to monitor for microbial products, barrier injury, and local cellular stress before deeper tissues become involved. Their strategic location allows them to become the first interpreters of environmental danger.
How do keratinocytes release early alarm signals during epidermal immune defense initiation?
Keratinocytes release early alarm signals during epidermal immune defense initiation by rapidly discharging preformed alarmins—endogenous molecules released from damaged cells to signal danger—such as IL-1α, and by producing additional cytokine signals that intensify local immune activation.
Preformed IL-1α can be released very rapidly after cellular injury or membrane disruption, which is why it is widely described as an alarmin that initiates early local inflammation before slower synthetic programs take over (PMC).
How does antimicrobial peptide production reinforce epidermal immune defense initiation?
Antimicrobial peptide (AMP) production—small proteins that directly kill or inhibit microorganisms—reinforces epidermal immune defense initiation by adding a rapid chemical layer of defense, deploying molecules such as LL-37 and defensins that can directly damage microbes and shape local inflammatory signaling.
AMPs matter because immune defense is not purely cellular; the epidermis also releases molecules that directly reduce microbial pressure. These peptides function as the skin’s own “natural antibiotics,” disrupting bacterial membranes on contact.
How does epidermal immune defense initiation differ between resident and recruited immune cells?
Epidermal immune defense initiation differs between resident and recruited immune cells in both timing and function, with resident cells providing near-immediate detection while recruited cells add larger-scale inflammatory reinforcement later.
| Immune Role | Resident Cells (Sentinels) | Recruited Cells (Reinforcement) |
|---|---|---|
| Timing | Immediate / Near-immediate | Delayed (hours to days) |
| Main Function | Surveillance and detection | Pathogen clearance and escalation |
| Functional Goal | Start the response | Help resolve the response |
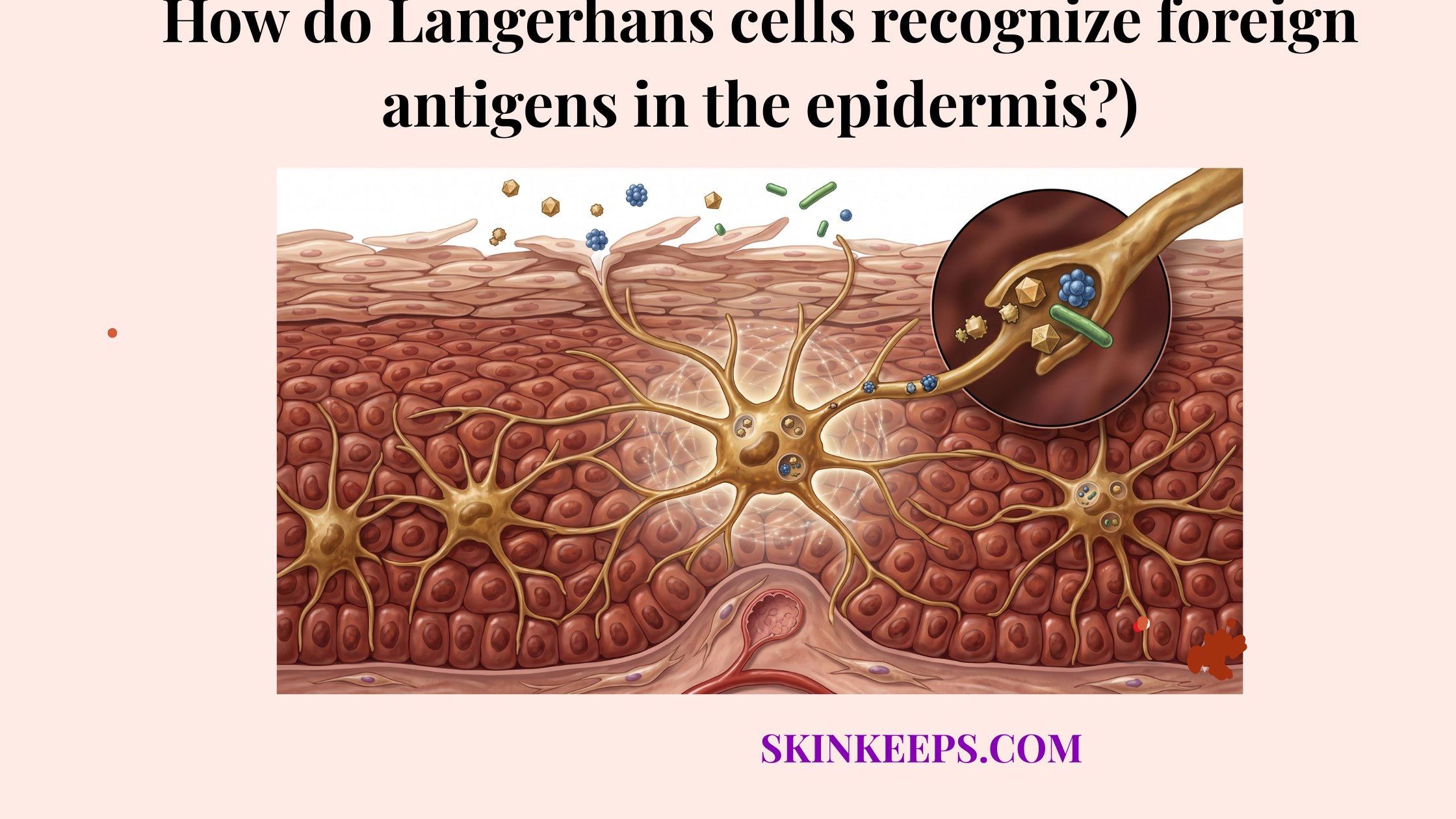
How does Langerhans cell migration connect epidermal immune defense initiation to adaptive immunity?
Langerhans cells—specialized resident immune cells that capture and process foreign antigens—migration connects epidermal immune defense initiation to adaptive immunity (the specialized long-term immune memory system) by capturing microbial antigens at the breach site and transporting that xog to regional lymph nodes to support later T-cell priming.
This bridging role depends on how Langerhans cells recognize foreign antigens in the epidermis, as they must process and internalize danger signals before moving. Langerhans cells upregulate CCR7 during migration toward skin-draining lymph nodes (PMC).
What pitfalls cause epidermal immune defense initiation to turn into chronic inflammation?
Pitfalls cause epidermal immune defense initiation to turn into chronic inflammation when the startup response fails to resolve and persistent barrier disruption keeps reactivating pro-inflammatory signaling.
In psoriasis, persistent activation of the IL-23/Th17 inflammatory axis is a central mechanism of chronic disease, and IL-23 is more accurately tied to dendritic-cell and innate immune circuit dysregulation than to a vague generic signal (PMC).
- False alarm → persistent barrier disruption keeps danger signaling active
- Resolution failure → resident and recruited pathways amplify instead of resolving
What are the key checkpoints in functional epidermal immune defense initiation?
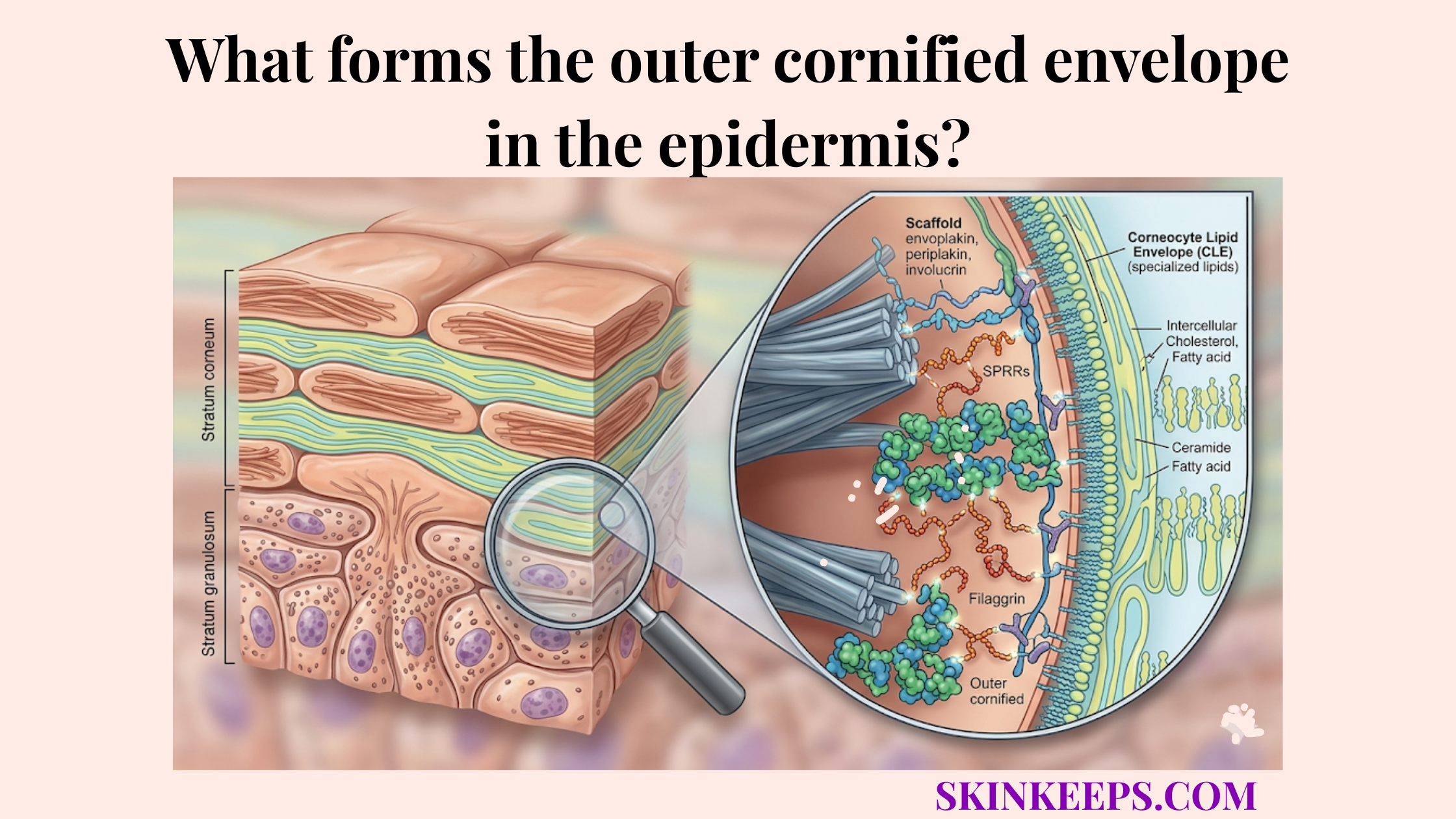
Functional epidermal immune defense initiation requires the successful execution of detection, alarmin release, antimicrobial reinforcement, and bridging from innate signaling to adaptive immune memory.
A functional epidermal immune response is not defined by inflammation alone. It is defined by fast detection, effective signaling, appropriate escalation, and controlled resolution.
Final Functional Checkpoints Checklist
Use this framework to evaluate epidermal immunity as a staged response system rather than a vague concept of skin defense.
Ultimately, when you master the intelligence of the epidermis, you master the health of the skin.