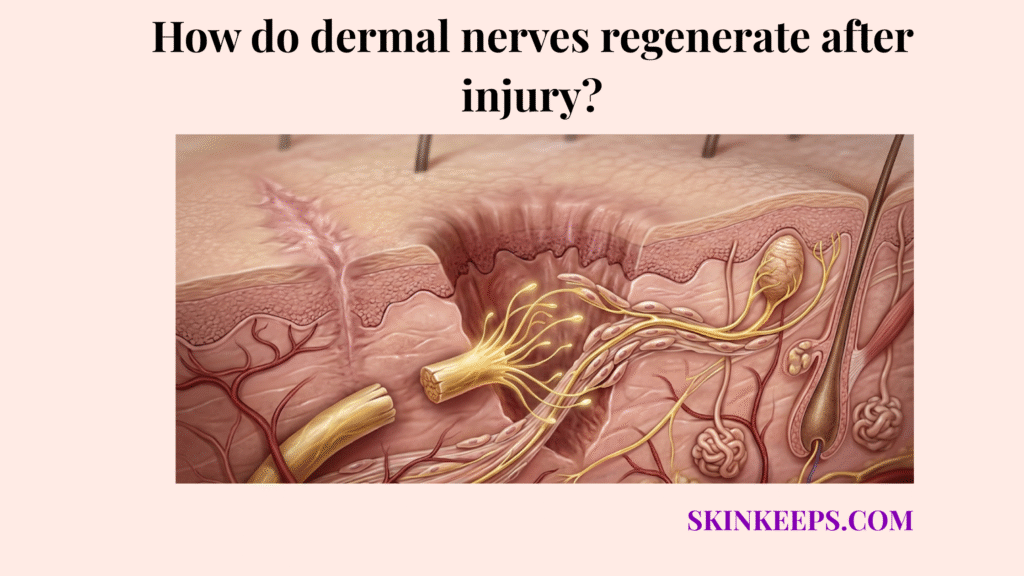
Dermal nerves regenerate after injury through a staged repair process in which the damaged distal segment is first broken down and cleared away, then guided regrowth begins from the surviving proximal nerve stump.
Patients are often confused when a laceration or deep scrape visibly closes in a week, yet the area remains completely numb or strangely painful for months. This happens because closing the physical skin layer is a rapid structural patch job, whereas peripheral nerve regeneration is a highly delicate, microscopic rewiring process.
Understanding how dermal nerves regenerate after injury requires tracking this slow biological progression: initial cleanup, Schwann-cell guidance pathway formation, growth cone advancement, measuring the odds of recovery versus failure, and identifying what supportive therapies actually optimize the repair environment.
What is the biological baseline for how dermal nerves regenerate after injury?
The biological baseline for how dermal nerves regenerate after injury is that meaningful regrowth cannot begin until the damaged distal nerve segment is dismantled and the repair environment is prepared for a new axon to advance.
When a nerve in the dermis is severed, it splits into two functional parts. The proximal stump is the segment still connected to the central nervous system; it remains alive and will eventually produce the new nerve sprout. The distal stump is the segment disconnected from the cell body; it loses its life support and must die.
This means the body cannot simply “glue” the two broken ends back together. The dead distal wire must be entirely hollowed out so the proximal wire can grow into the empty tube.
StatPearls notes that Wallerian degeneration occurs in the distal axon stump within about 24 to 36 hours after peripheral nerve injury, which gives the clearest quantitative anchor for the first biological shift after injury (Cruz and Townsend, 2023 — NCBI).
How does Wallerian degeneration facilitate how dermal nerves regenerate after injury?
Wallerian degeneration facilitates how dermal nerves regenerate after injury by clearing the damaged distal axon and myelin so that new growth is not blocked by structural debris.
Despite sounding like a complication, Wallerian degeneration is a highly productive, controlled cleanup phase. The disconnected distal nerve breaks itself down into fragments.
Following this breakdown, resident cells and recruited macrophages (immune cells specialized in phagocytosis) swarm the area to aggressively consume the dead axon and any residual myelin debris. If this debris is not cleared, the physical and chemical roadblocks will permanently stall any regeneration from the proximal side. This intense cleanup is an essential prerequisite before normal dermal function can be restored.
How do Schwann cells support how dermal nerves regenerate after injury?
Schwann cells support how dermal nerves regenerate after injury by converting into a repair phenotype, aligning along the damaged pathway, and creating guidance structures that direct new axonal growth.
Typically, Schwann cells act as insulators for peripheral nerves. However, upon injury, they shed their myelin-producing identity and dedifferentiate into specialized repair Schwann cells. They assist macrophages in consuming debris and then stretch out end-to-end to form microscopic biological tunnels.
These organized cellular columns, called Büngner bands, sit inside hollow basal lamina tubes left behind by the dead nerve. These bands physically and chemically point the way, ensuring the new nerve fiber does not wander aimlessly into surrounding dermal tissue.
How do growth cones dictate how dermal nerves regenerate after injury?
Growth cones dictate how dermal nerves regenerate after injury by acting as the searching and advancing front of the regenerating axon, sensing guidance cues and moving through Schwann-cell pathways toward reconnection.
Once the path is clear, the surviving proximal stump initiates axonal sprouting. The tip of this new sprout swells into a highly sensitive, mobile structure known as a growth cone. The growth cone extends tiny finger-like projections (filopodia) that “taste” the tissue environment.
It searches for neurotrophic guidance cues—chemical signals released by the repair Schwann cells—that instruct it where to grow. Correct navigation by the growth cone matters just as much as the raw volume of regrowth; if the cone is misdirected, sensation will not return to the correct area of the skin.
How does the regeneration rate shape how dermal nerves regenerate after injury?
The regeneration rate shapes how dermal nerves regenerate after injury because even successful axonal regrowth is biologically slow, so sensory return often lags far behind visible skin healing.
Patients frequently assume that once a laceration closes and the scab falls off, the tissue is entirely healed. However, while epidermal cells renew in weeks, peripheral nerves regenerate at a notoriously slow pace. If you are wondering what is the typical regeneration rate of dermal nerve fibers, StatPearls states that peripheral axonal growth often follows a rough clinical rule of about 1 mm per day (Althagafi and Nadi, 2023 — NCBI). This means a nerve severed just a few centimeters below the skin surface can take months to successfully navigate the distance and restore sensation to the epidermis.
How does successful recovery compare with failed outcomes in how dermal nerves regenerate after injury?
Successful recovery and failed outcomes differ because dermal nerves regenerate after injury only when new axonal growth is guided cleanly enough to reconnect with an appropriate target. When patients ask, can full sensory function return after dermal nerve damage?, the answer depends heavily on whether the regenerating axon finds its original receptor.
Functional reinnervation (successful connection) results in the gradual return of sensory awareness. However, recognizing which factors slow dermal nerve recovery helps explain why severe trauma or heavy scar tissue can cause the growth cone to wander. This causes misdirected regeneration, where the nerve connects to the wrong target, or failed regeneration, resulting in chronic denervation and permanent numbness.
In severe cases, the trapped growth cone spirals into a disorganized, highly sensitive knot of nerve endings known as a neuroma, which generates painful, hypersensitive nerve firing instead of useful sensation.
| Recovery outcome | Structural result | Sensory outcome | Main problem |
|---|---|---|---|
| Successful regeneration | Axon reconnects through guidance path | Sensation gradually returns | Regrowth reached correct target |
| Failed or misdirected regeneration | Regrowth is blocked, misrouted, or incomplete | Persistent numbness, altered sensation, or pain | Pathway failure or incorrect reconnection |
| Neuroma formation | Chaotic nerve-end growth at injury site | Painful hypersensitivity or abnormal firing | Regrowth did not reconnect properly |
What specific therapies support how dermal nerves regenerate after injury?
The therapies most likely to support how dermal nerves regenerate after injury are those that improve the repair environment, metabolic support, and guidance conditions rather than pretending to create instant nerve recovery.
How do B-vitamins and metabolic support help how dermal nerves regenerate after injury?
B-vitamins and broader metabolic support help how dermal nerves regenerate after injury by supporting neuronal metabolism, myelin biology, and the biochemical environment needed for axonal repair.
Vitamin B12 acts as a crucial co-factor for myelin synthesis and nerve cell metabolism. Proper nutritional status ensures that the high-energy demands of the advancing growth cone are met. While Vitamin B6 also provides supportive value, megadosing is not a miracle cure; rather, maintaining excellent baseline metabolic health removes barriers to the body’s natural repair sequence.
How does Alpha-Lipoic Acid help support how dermal nerves regenerate after injury?
Alpha-Lipoic Acid may help support how dermal nerves regenerate after injury mainly by improving oxidative-stress conditions and neuropathic symptom biology rather than by acting as a proven stand-alone regeneration trigger.
As a potent antioxidant, it helps neutralize reactive oxygen species that accumulate around injured tissue, creating a less hostile environment for the fragile sprouting axon. It is widely recognized for supporting symptomatic relief in neuropathic conditions, stabilizing the environment so the nerve can focus energy on structural regrowth.
How does electrical stimulation help support how dermal nerves regenerate after injury?
Electrical stimulation may help support how dermal nerves regenerate after injury by enhancing the regenerative environment and, in some clinical settings, accelerating axon growth and sensorimotor recovery.
Targeted, clinically administered low-frequency electrical stimulation applied shortly after injury has been shown to upregulate neurotrophic factors and accelerate axonal crossing of the injury gap. It acts as structured physical therapy for the nerve, promoting faster alignment and directional growth when utilized properly.
Therapeutic Support Framework
- Problem → nerve regrowth is slow and vulnerable to failure.
- Implication → sensory recovery may stall or reconnect poorly.
- Solution → support metabolic health, preserve repair conditions, and use structured therapy where appropriate.
What are the summary facts for how dermal nerves regenerate after injury?
The summary facts are that dermal nerves regenerate after injury only after distal cleanup occurs, Schwann-cell guidance remains available, and slow axonal regrowth successfully reconnects with the correct target.
Nerve Regeneration Summary Checklist
What steps can you take to optimize how your dermal nerves regenerate after injury?
Dermal nerve recovery is optimized when patients support general nerve health, protect healing tissues, and seek structured evaluation when sensory return stalls or becomes abnormal.
Recovery Optimization Checklist
Quick Answers About How Dermal Nerves Regenerate After Injury
What happens first when a dermal nerve is injured?
When a dermal nerve is injured, the first step is the breakdown of the disconnected nerve segment. This controlled self-destruction must occur before dermal nerves regenerate after injury, ensuring the physical path is completely clear for new axonal sprouting.
What is Wallerian degeneration?
Wallerian degeneration is the active, programmed clearance of the damaged distal stump and its surrounding myelin debris. This cleanup phase removes physical and chemical roadblocks so that dermal nerves regenerate after injury without obstruction.
What do Schwann cells do after nerve injury?
Schwann cells reprogram into a specialized repair state, helping macrophages consume debris and then forming aligned guidance tunnels (Büngner bands). They essentially build the biological highway that dermal nerves regenerate after injury through.
How fast do dermal nerves regrow?
Dermal nerves regenerate after injury at a notoriously slow biological pace, typically advancing at approximately 1 millimeter per day. This speed heavily depends on the injury severity, metabolic health, and the integrity of the guidance pathways.
Why does skin heal before sensation returns?
Skin heals before sensation returns because epidermal cell turnover and wound closure occur in weeks, while axonal regrowth takes months. Dermal nerves regenerate after injury much slower because they must rebuild microscopic connections millimeter by millimeter.
What is a growth cone in nerve repair?
A growth cone is the highly sensitive, advancing tip of a sprouting axon. It acts as the navigation center when dermal nerves regenerate after injury, physically pulling the nerve forward by sensing neurotrophic guidance cues in the tissue.
Can failed nerve regeneration cause a neuroma?
Yes. When dermal nerves regenerate after injury but fail to find a clear path, the axons can become trapped and form a disorganized, painful knot of nerve endings known as a neuroma.
Does electrical stimulation help nerve recovery?
Electrical stimulation can help create a more favorable environment for nerve repair. While it does not act as a magical cure, clinical application early in the process supports how dermal nerves regenerate after injury by enhancing metabolic signaling.
Conclusion
In conclusion, dermal nerves regenerate after injury through a slow but organized biological sequence in which degeneration clears the path, Schwann cells guide new growth, and successful reconnection determines whether meaningful sensation returns.
Because the body prioritizes structural skin closure long before the complex neurological wiring can be rebuilt, sensory return naturally lags. Patience, systemic metabolic support, and clinical monitoring for signs of misdirected repair remain the most pragmatic ways to support functional nerve recovery.