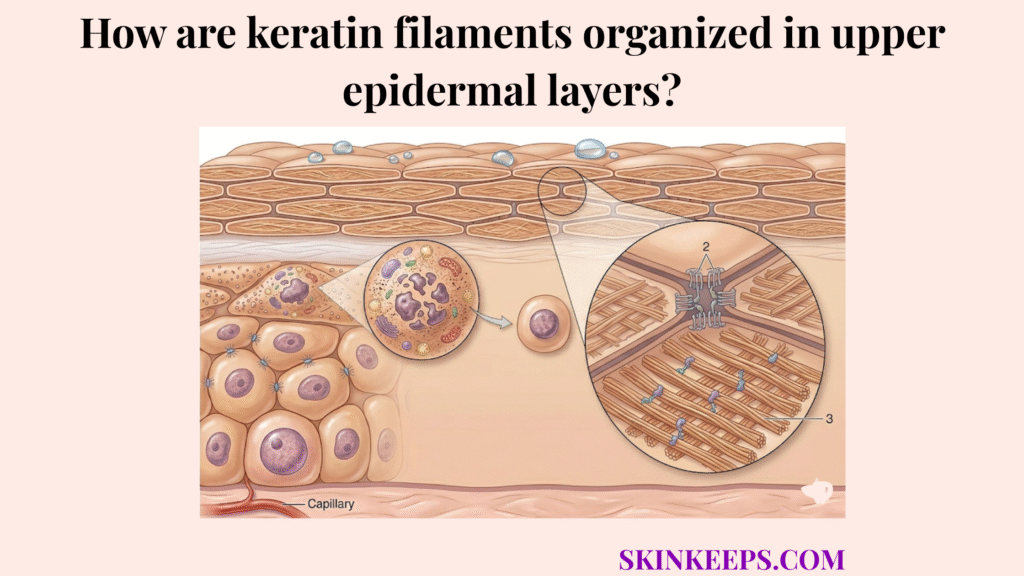
Keratin filaments are organized in upper epidermal layers through a progressive structural transition in which suprabasal keratinocytes, the cells positioned above the basal layer, develop denser bundled filament networks that become more peripherally anchored and finally compact into the corneocyte matrix. This organization changes step by step as keratinocytes move upward, shifting from flexible intracellular scaffolding toward a denser and more mechanically committed architecture.
Keratin filament organization is not random intracellular clutter but a highly ordered structural system that gives the epidermis tensile strength, tissue continuity, and resistance to mechanical stress. Intermediate filaments are about 10 nm in diameter, which is why consistent filament organization matters so much mechanically. (ncbi.nlm.nih.gov)
Understanding how keratin filaments are organized in upper epidermal layers makes it easier to interpret normal epidermal maturation, distinguish organized from disrupted cornification, and connect microscopic structure to visible barrier quality.
Why do keratin filaments need organized architecture in upper epidermal layers?
Keratin filaments need organized architecture in upper epidermal layers because epidermal mechanical resilience depends on how those structural proteins are arranged, reinforced, and integrated across differentiating keratinocytes.
The epidermis depends on internal structural order because the same proteins can produce very different outcomes if they are poorly arranged. Disordered networks fail to distribute mechanical loads, leading to tissue fragility even if protein levels are normal.
This arrangement functions as the physical foundation of the skin’s defense: the barrier is strong not only because keratin exists, but because keratin is organized into a mechanically useful network.
| Structural condition | Barrier consequence |
|---|---|
| Ordered filament network | Better tensile resilience and tissue continuity |
| Poorly integrated network | Higher fragility under mechanical stress |
| Dense terminal compaction | Stronger surface-cell architecture |
How does keratin filament organization shift across the epidermal strata?
Keratin filament organization shifts across the epidermal strata by becoming progressively denser and more aggregated as keratinocytes move from early suprabasal bundling into granular-layer condensation and final compaction in the stratum corneum.
Upper-layer organization is not a static snapshot but a directional sequence from looser filament networks to more condensed structural states. The upper epidermis moves from the spinous layer, into the stratum granulosum, the late-living granular layer, and finally into the stratum corneum, the outer cornified layer.
During this migration, cells develop tonofilaments, which are bundled keratin intermediate filaments inside keratinocytes. These bundles provide the requisite rigidity for the rising tissue.
Looser upper-living networks become progressively more committed and compact as terminal differentiation advances. This directional shift ensures that cells arriving at the surface are fully “armored” for barrier duty.
Key takeaway: Keratin filament organization shifts by becoming more structurally committed as cells move upward.
How do K1 and K10 stabilize keratin filament organization?
K1 and K10 stabilize keratin filament organization by replacing the basal K5/K14 pair during differentiation, creating a suprabasal network better suited to the tensile demands of the upper epidermis.
In this biological switch, K5/K14 acts as the basal keratin pair associated with proliferative basal cells, while K1/K10 functions as the suprabasal keratin pair associated with structural reinforcement during differentiation.
This replacement marks a functional shift from proliferative support in basal cells to structural reinforcement in differentiating suprabasal cells. Without this switch, the cell would lack the mechanical rigidity required to form a protective shield.
Keratin intermediate filaments maintain a diameter of about 10 nm, and the suprabasal K1/K10 switch helps preserve this mechanically useful filament architecture as differentiation progresses (ncbi.nlm.nih.gov).
| Epidermal zone | Dominant keratin pair | Main functional emphasis |
|---|---|---|
| Basal layer | K5/K14 | Proliferative support and basal resilience |
| Suprabasal layer | K1/K10 | Structural stabilization and tensile support |
| Functional shift | Reinforcement | From mitotic support to architectural strength |
How does filaggrin reorganize keratin filaments into the macrofibrillar matrix?
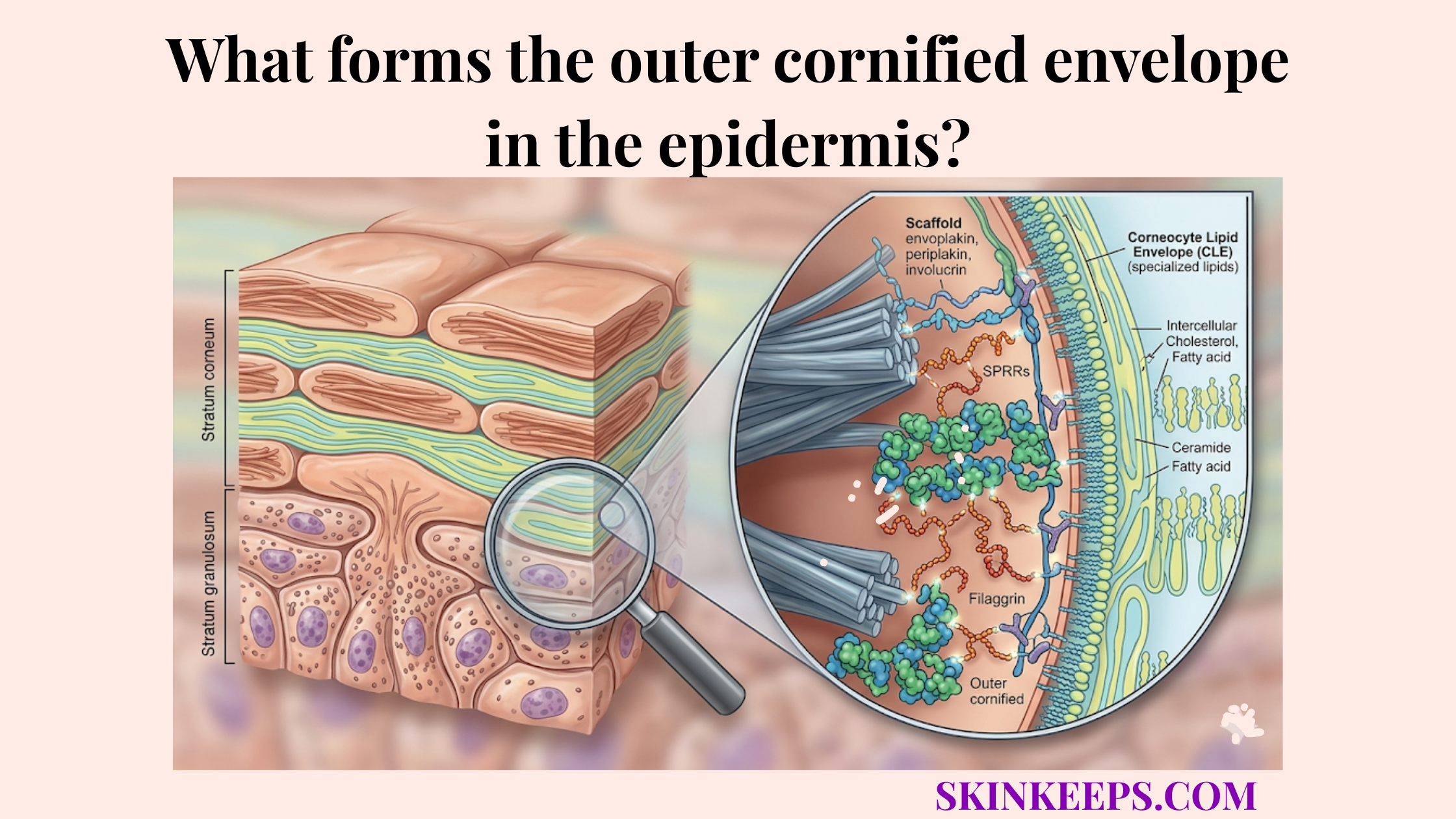
Filaggrin reorganizes keratin filaments by promoting the lateral aggregation of keratin bundles, collapsing the more open cytoskeletal network into the dense macrofibrillar matrix that characterizes the mature corneocyte.
In this condensation phase, filaggrin acts as a specialized keratin-bundling protein that condenses the filament network into an insoluble composite. This creates the macrofibrillar matrix, the dense compacted keratin architecture of the mature corneocyte.
This is the stage where open intracellular scaffolding becomes a denser and flatter terminal structure. Filaggrin was named for its ability to aggregate keratin intermediate filaments, and that role remains central to terminal differentiation (pmc.ncbi.nlm.nih.gov).
How does keratin filament organization differ between the stratum granulosum and stratum corneum?
Keratin filament organization differs between the stratum granulosum and stratum corneum because the granular layer is still actively reorganizing the network through maturation machinery, whereas the corneum reflects the final, irreversible state of structural compaction.
In this anatomical transition, the stratum granulosum acts as the late-living granular layer where terminal reorganization intensifies, while the stratum corneum represents the outer cornified layer composed of fully matured surface cells.
This comparison highlights organization in progress versus organization completed. In the granular layer, filaments are being bundled; in the corneum, they are part of a solid, waterproof protein-lipid matrix.
| Layer | Cell state | Keratin filament organization | Structural result |
|---|---|---|---|
| Stratum granulosum | Living (late) | Dense but actively reorganizing | Transitional hardening |
| Stratum corneum | Fully cornified | Highly compacted keratin matrix | Final protective barrier |
How do desmosomes anchor keratin filaments to the cell periphery?
Desmosomes anchor keratin filaments to the cell periphery by serving as junctional attachment sites that convert individually strong cells into a mechanically integrated epidermal sheet.
In this model, desmosomes function as specialized cell-cell junctions that anchor keratin filaments at the cell edge. To understand this tissue-wide order, compare keratin filaments to steel rebar and desmosomes to the welds that join those reinforcing bars across the structure.
Filament strength alone is not enough; tissue continuity requires peripheral anchoring between cells. Desmosomes anchor the intermediate filament cytoskeleton to the plasma membrane and are central to epithelial mechanics, ensuring the tissue remains cohesive (f1000research.com).
What misconceptions commonly distort how keratin filament organization is understood during cornification?
Common misconceptions distort keratin filament organization by implying that filaments clump randomly during cornification, when the upper epidermis actually follows an ordered program of structural maturation and condensation.
To correct this, we must define cornification as the final epidermal differentiation program that produces the mature cornified surface cell. It is not a chaotic collapse, but a precisely timed biological reorganization.
Misconception Correction Grid
How can barrier weaknesses be traced back to disruptions in keratin filament organization?
Barrier weaknesses can be traced back to disruptions in keratin filament organization when faulty keratin pairing, poor filaggrin aggregation, or unstable anchoring creates a fragile cell architecture that cannot resist mechanical shear effectively.
When filaggrin aggregation fails, it means that keratin bundles are failing to condense into the denser terminal structure they need for barrier duty. This leads to porous, weak “bricks” that allow barrier disruption and increased water loss.
Mutations in epidermal keratin genes provide direct evidence that disrupted keratin architecture leads to cell fragility, blistering, and hyperkeratotic disease, showing that filament organization is a core driver of mechanical barrier strength (ncbi.nlm.nih.gov).
| Structural state | Mechanical consequence | Clinical expression |
|---|---|---|
| Normal filament organization | Strong barrier mechanics | Resilient epidermal sheet |
| Disrupted keratin architecture | Weaker cell architecture | Fragility, scaling, flaking |
| Failed anchoring | Loss of tissue integration | Poor resistance to shear stress |
What does successful keratin filament organization require?
Successful keratin filament organization requires the correct suprabasal keratin switch, effective filaggrin-mediated aggregation, and stable desmosomal anchoring so the epidermis can achieve true mechanical resilience.
A strong model of keratin filament organization should explain not just where the filaments are, but how they change, what stabilizes them, what anchors them, and what final structure they produce.
Final Filament-Organization Validation Checklist
Use this framework to evaluate epidermal maturation as a structural sequence rather than a list of isolated proteins or layers.
Ultimately, when you master the architecture of the epidermis, you master the health of the skin.