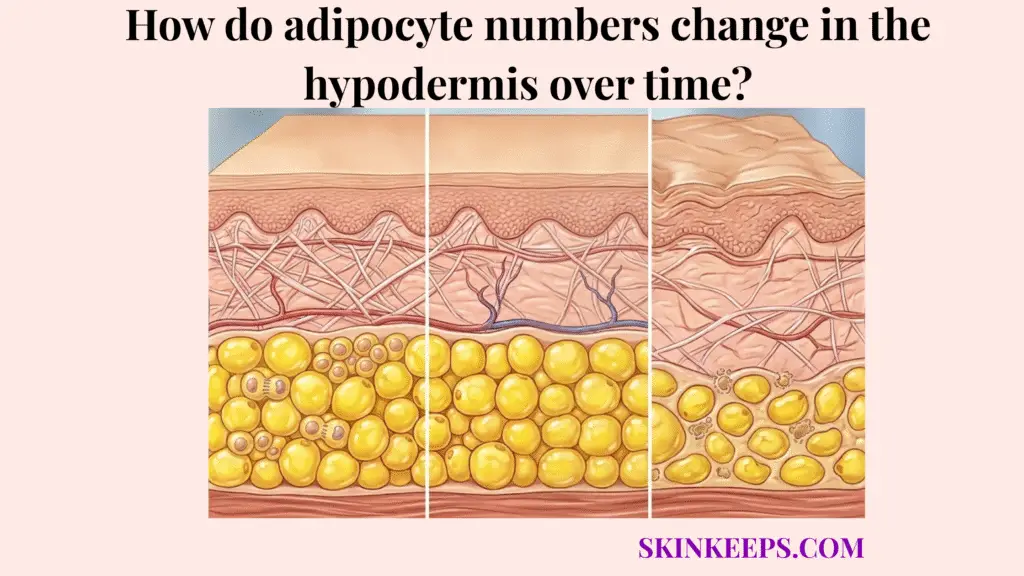
Adipocyte numbers in the hypodermis change over time because adult fat cells normally turn over slowly, but aging weakens the balance between cell death and cell replacement, making the tissue more vulnerable to a gradual net loss of adipocyte number. Throughout early adulthood, the deep support layer maintains a state of relative equilibrium. However, as the intrinsic cellular biology matures, the mechanisms responsible for replenishing this critical structural padding begin to fail, resulting in a quantifiable decline in the absolute cell count residing within the subcutaneous matrix.
Understanding this trajectory requires a strict distinction between cell number and cell size. When an individual achieves ordinary weight loss through a caloric deficit, the existing adipocytes merely shrink and release their stored lipids while remaining biologically alive and anchored in the tissue. In stark contrast, aging drives an entirely different mechanism: it actively removes the cells through apoptosis while simultaneously stripping the tissue of its ability to regenerate them. This fundamental failure in replacement is what permanently alters the total adipocyte population in the aging hypodermis.
What exactly happens to the total adipocyte population in the hypodermis as we age?
As the hypodermis ages, the total adipocyte population becomes more vulnerable to a net negative turnover balance, where replacement no longer keeps pace with loss as reliably as it did in younger adult tissue. Cellular turnover is a continuous, tightly regulated process essential for maintaining the integrity of subcutaneous adipose tissue. Adipocytes are exceptionally long-lived entities, but they are not strictly permanent; they follow a programmed lifecycle requiring meticulous replacement to ensure the deep tissue maintains its structural volume over decades.
During a normal cellular lifespan, older or damaged adipocytes eventually undergo apoptosis, a highly orchestrated sequence of programmed cell death. In a robust, youthful biological environment, resident adipose progenitor cells immediately differentiate into new, fully functional adipocytes to fill the resulting void. However, as aging progresses, the efficiency of this entire regenerative side of the equation rapidly deteriorates, rendering the replacement process increasingly undependable.
Consequently, the hypodermal environment enters a state of definitive “net math” decline. If mature cells continue to die at a baseline rate, but significantly fewer preadipocytes successfully mature to take their place, the long-term biological trajectory permanently shifts toward a lower total cell number. Adult human adipocytes turn over at approximately 10% per year, keeping the population relatively stable under healthy baseline conditions; however, when aging disrupts this steady 10% replacement rate, profound structural depletion inevitably follows (Arner et al., 2019) [PMC].
Why does the biological renewal of hypodermal fat cells slow down over time?
The biological renewal of hypodermal fat cells slows down over time because aging reduces adipose progenitor competence, increases senescence-related signaling, and weakens the adipogenic environment needed to replace dying adipocytes. The root of this deceleration lies deeply within stem cell exhaustion. Within the hypodermis, a specific pool of preadipocytes and adipose-derived progenitors exist solely to proliferate and mature upon demand; however, progressive aging fundamentally blunts their ability to divide and differentiate normally into viable, lipid-storing cells.
Compounding this progenitor failure is the mounting burden of cellular senescence throughout the adipose layer. As surrounding mature cells and supportive fibroblasts become senescent, they cease their normal physiological duties and begin secreting a toxic blend of inflammatory cytokines and destructive matrix enzymes known as the Senescence-Associated Secretory Phenotype (SASP). This highly inflammatory signaling explicitly suppresses the adipogenic behavior of neighboring healthy cells, actively shutting down local regenerative efforts.
Crucially, this determines biologic reversibility. Size loss induced by dieting is highly reversible; introducing a caloric surplus will rapidly trigger cellular hypertrophy, re-expanding those living cells to restore fullness. However, age-related number loss is fundamentally resistant to simple nutritional reversal. Because aging includes an intrinsically impaired renewal biology, losing the baseline 10% annual turnover means those missing cells cannot be coaxed back into existence merely by eating more (Arner et al., 2019) [PubMed].
| Biological Process | Youth / younger adult baseline | Aging pattern | Net Cellular Effect |
|---|---|---|---|
| Adipocyte turnover | Slow but ongoing | Less well balanced | Higher risk of deficit |
| Preadipocyte differentiation | More competent | Impaired | Fewer replacements |
| Senescent signaling | Lower burden | Higher burden | Adipogenesis suppression |
| Tissue homeostasis | More stable | More dysregulated | Number maintenance worsens |
How does a shrinking fat cell count differ from a decrease in fat cell size?
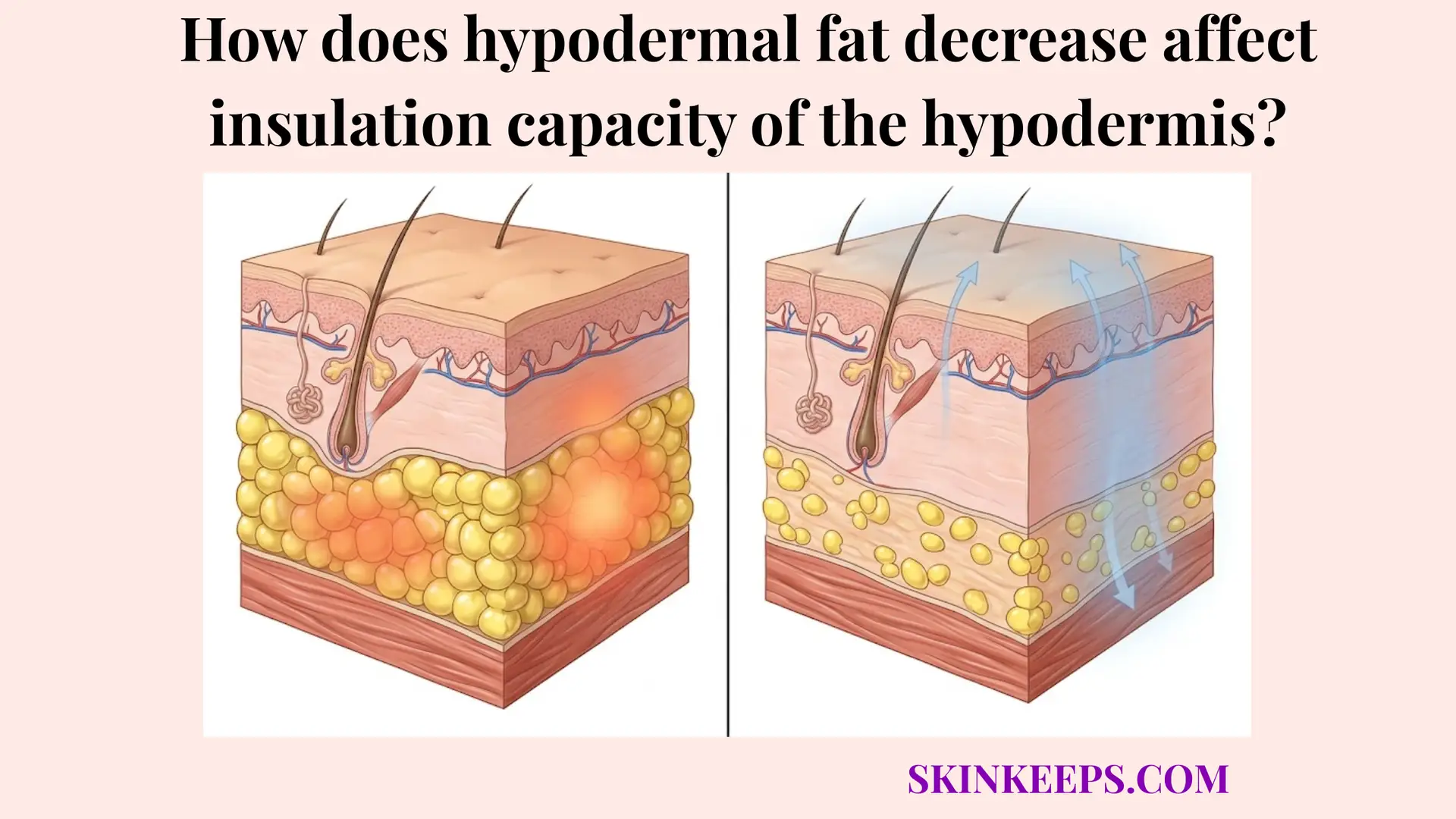
A shrinking fat cell count differs from a decrease in fat cell size because weight loss usually reduces lipid content inside living adipocytes, while aging-related number loss removes or fails to replace the adipocytes themselves. This critical nuance separates transient nutritional changes from permanent physiological aging. During periods of caloric deficit or intense metabolic expenditure, a healthy living adipocyte undergoes atrophy; its internal lipid vacuole empties, causing the cell to shrink dramatically in diameter, yet the cellular membrane and vital nucleus remain fully intact.
Conversely, a shrinking cell count signifies a structural deletion. When adipocytes undergo apoptosis and are not adequately replaced by the dysfunctional progenitor pool, the hypodermis is literally losing functioning cellular units, not merely dispensing stored energy content. This depletion physically hollows out the tissue scaffold, permanently altering the volume capacity of the deep fat compartments regardless of the patient’s current daily caloric intake.
Crucially, this determines biologic reversibility. Size loss induced by dieting is highly reversible; introducing a caloric surplus will rapidly trigger cellular hypertrophy, re-expanding those living cells to restore fullness. However, age-related number loss is fundamentally resistant to simple nutritional reversal. Because aging includes an intrinsically impaired renewal biology, losing the baseline 10% annual turnover means those missing cells cannot be coaxed back into existence merely by eating more (Arner et al., 2019) [PMC].
| Feature | Shrinking Size (Weight Loss) | Shrinking Number (Aging) |
|---|---|---|
| Main change | Less lipid inside the cell | Fewer actual adipocytes |
| Different biologic logic | Cell survives? Usually yes | Not necessarily |
| Reversibility | Size change is more reversible | Aging + impaired renewal |
| Main driver | Energy deficit | Tissue aging is harder to reverse |
| Restoring fullness | Refeeding can enlarge cells | Lost cells are not easily regenerated |
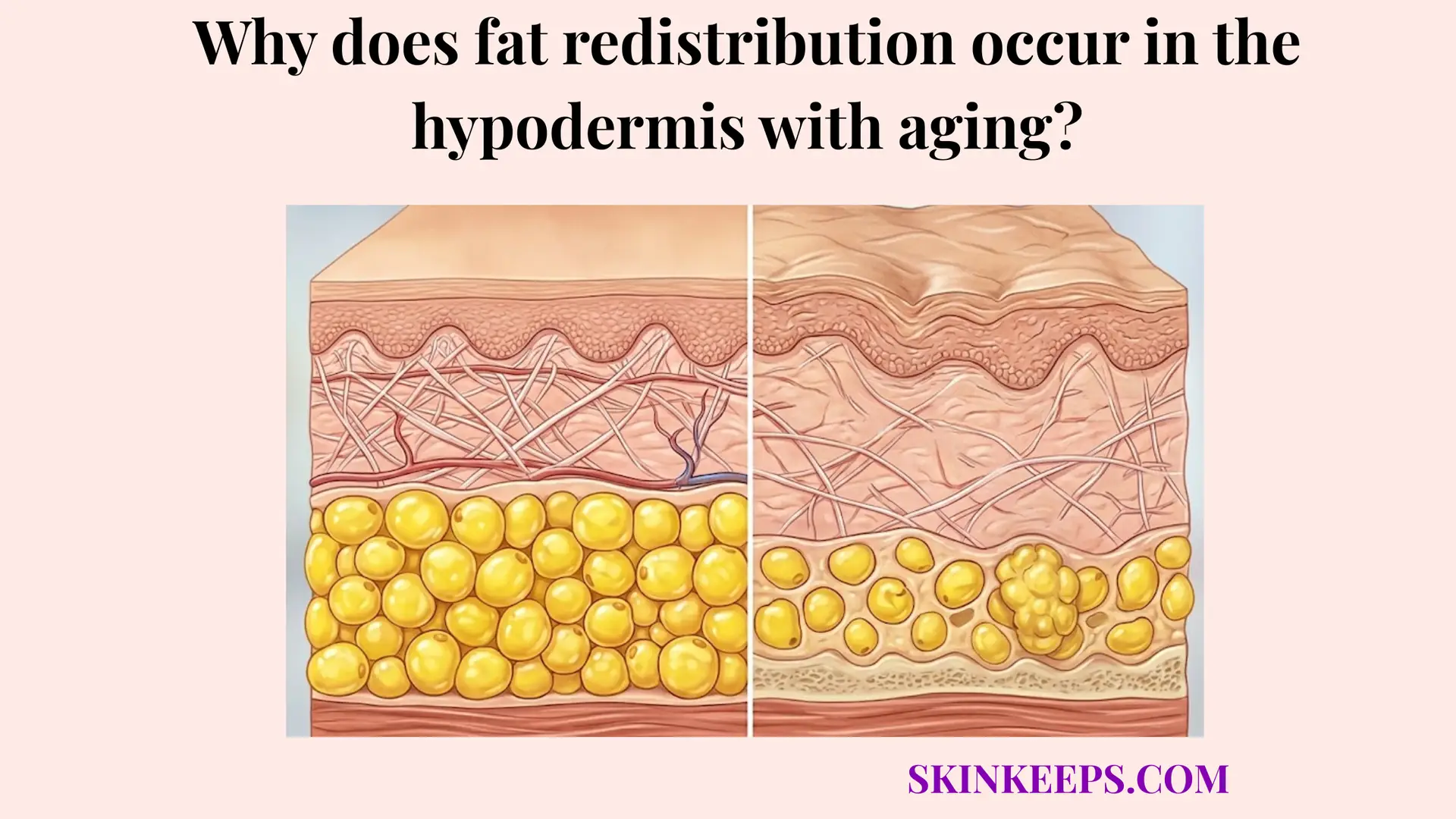
At what life stages does the loss of subcutaneous adipocytes accelerate most rapidly?
Loss of subcutaneous adipocytes should be framed as a progressive age-related shift, not as a single universal age cliff, although clinical acceleration becomes more plausible from midlife onward as senescence, hormonal change, and regenerative decline accumulate. During the 20s and 30s, the hypodermis relies on a robust progenitor pool. Throughout these earlier decades, adult adipocyte turnover is much more likely to remain closer to strict equilibrium, wherein apoptosis is effectively matched by newly generated fat cells, preserving baseline structural fullness.
As patients transition into their 40s and 50s, this phase marks a critical biological threshold where midlife hormonal and regenerative changes radically alter the cellular equation. The systemic decline in protective hormones, such as estrogen, severely weakens connective tissue integrity while simultaneously exacerbating progenitor exhaustion. It is during this window that the net negative turnover balance often shifts from a silent microscopic event into a visible, clinically significant anatomical deflation.
Advancing into the 60s and beyond, the accumulated burden of advanced senescence and drastically weakened cellular replacement make these long-standing deficits unmistakably obvious. By this stage, the hypodermis has likely operated under a replacement deficit for years, manifesting as profound facial skeletalization and tissue laxity. Rather than assigning an arbitrary percentage to an exact age, it is vital to recognize that when the baseline 10% annual turnover fails sequentially across decades, structural collapse is inevitable (Arner et al., 2019) [PMC].
Which clinical procedures actually replace a depleted subcutaneous fat cell population?
The clinical procedure that most directly replaces a depleted subcutaneous fat cell population is autologous fat grafting, while fillers mainly add space-occupying volume and biostimulators mainly improve matrix support rather than creating a new native adipocyte population. For patients demanding true cellular restoration, autologous fat grafting stands alone. This surgical technique physically harvests living adipose tissue—rich with mature adipocytes and viable adipose-derived stem cells—and transplants it, directly populating the hollowed hypodermis with living biology.
In contrast, hyaluronic acid (HA) fillers must be distinctly understood as powerful volumizing materials, not cellular replacement therapies. HA injections excel at instantly simulating structural volume and refining facial contours by attracting and binding water molecules deep within the tissue layers. However, they are composed of inert, sterile gels; they deposit absolutely no living cells and exert no direct influence on replacing the vanished adipocyte population.
Similarly, biostimulators like Poly-L-lactic acid (PLLA) function through an entirely different physiological axis. These agents act as matrix-support strategies, intentionally provoking a mild inflammatory response to force surrounding fibroblasts into producing dense new collagen networks. While this expertly thickens the dermal and subdermal scaffolding to tighten lax tissue, it does not instruct the body to generate a new native adipocyte population (Schipper et al., 2022) [Europe PMC].
| Treatment | Adds New Cells? | Mechanism of Action | Longevity |
|---|---|---|---|
| Autologous fat grafting | Yes, by transferring living adipose tissue | Structural cell transfer | Variable, technique-dependent |
| Hyaluronic acid filler | No | Space-occupying volume simulation | Temporary |
| Biostimulator (e.g. PLLA) | No direct adipocyte replacement | Collagen / matrix stimulation | Gradual and longer-lasting than simple filler, but not cell replacement |
Which metabolic stressors accelerate the premature death of your hypodermal adipocytes?
Metabolic and environmental stressors can accelerate premature adipocyte injury and loss by increasing oxidative stress, damaging progenitor function, and making the adipose niche less supportive of long-term cell survival. Chronic oxidative stress remains a primary instigator of cellular destruction within the deep tissue. For instance, the heavy systemic toxic load induced by chronic smoking drastically starves the hypodermal environment of optimal oxygenation, generating massive free-radical activity that severely injures the delicate lipid membranes of living adipocytes.
Additionally, severe ultraviolet (UV) radiation triggers cascading inflammatory stress signals that permeate far beneath the superficial epidermis. This aggressive photo-induced oxidative stress heavily disrupts the local metabolic microenvironment. When the adipose niche is subjected to sustained inflammatory cytokines triggered by UV damage, it actively suppresses healthy adipogenic behavior, ensuring that fewer progenitor cells successfully mature to execute their replacement duties.
Finally, the accumulation of Advanced Glycation End Products (AGEs)—driven by chronic high blood sugar and poor metabolic stability—acts as a devastating molecular wrecking ball. Glycation stress structurally hardens the connective matrix surrounding the cells and directly signals for cellular deletion. Strikingly, specific mechanistic studies have demonstrated that AGEs actively promote apoptosis in human adipose-derived stem cells through targeted receptor pathways, literally forcing the premature death of the very cells needed for hypodermal renewal (Wang et al., 2015) [Springer].
What is the ultimate biological checklist for preserving your remaining hypodermal cell count?
The ultimate biological checklist for preserving your remaining hypodermal cell count is to protect adipocyte turnover from avoidable stress, stabilize metabolic health, avoid repeated tissue-damaging cycles, and understand that once true cell number is lost, ordinary diet alone does not simply rebuild it. Because aging naturally impairs the body’s regenerative toolkit, patients must act defensively to shield their existing cell population. Stabilizing metabolic health prevents the aggressive glycation and oxidative damage that prematurely triggers adipocyte apoptosis.
It is crucial to recognize the rigid limits of nutritional intervention regarding cellular architecture. While maintaining a stable caloric intake prevents the existing adipocytes from continuously shrinking and expanding—a mechanical stress that weakens the surrounding connective tissue—diet alone cannot spawn new cells. Once the baseline 10% annual adipocyte turnover is compromised and structural depletion has occurred, consuming excess calories will only engorge the remaining cells without replacing the deleted population.
If profound structural restoration of cell number becomes an aesthetic necessity, the patient must match the treatment to the biology. Understanding that fillers and biostimulators offer magnificent, yet simulated, matrix support is vital. When the true clinical objective is the direct addition of living cellular units to replace a severely diminished hypodermal population, autologous fat grafting remains the only anatomically accurate intervention discussed (Arner et al., 2019) [PMC].
Adipocyte Population Preservation Checklist
Frequently Asked Questions About Adipocyte Aging
Are adipocytes permanent cells in the hypodermis?
No. Adipocytes are long-lived but not entirely permanent. In a healthy adult, they undergo a continuous, slow turnover process where old or damaged cells undergo apoptosis and are replaced by newly differentiated preadipocytes to maintain tissue homeostasis.
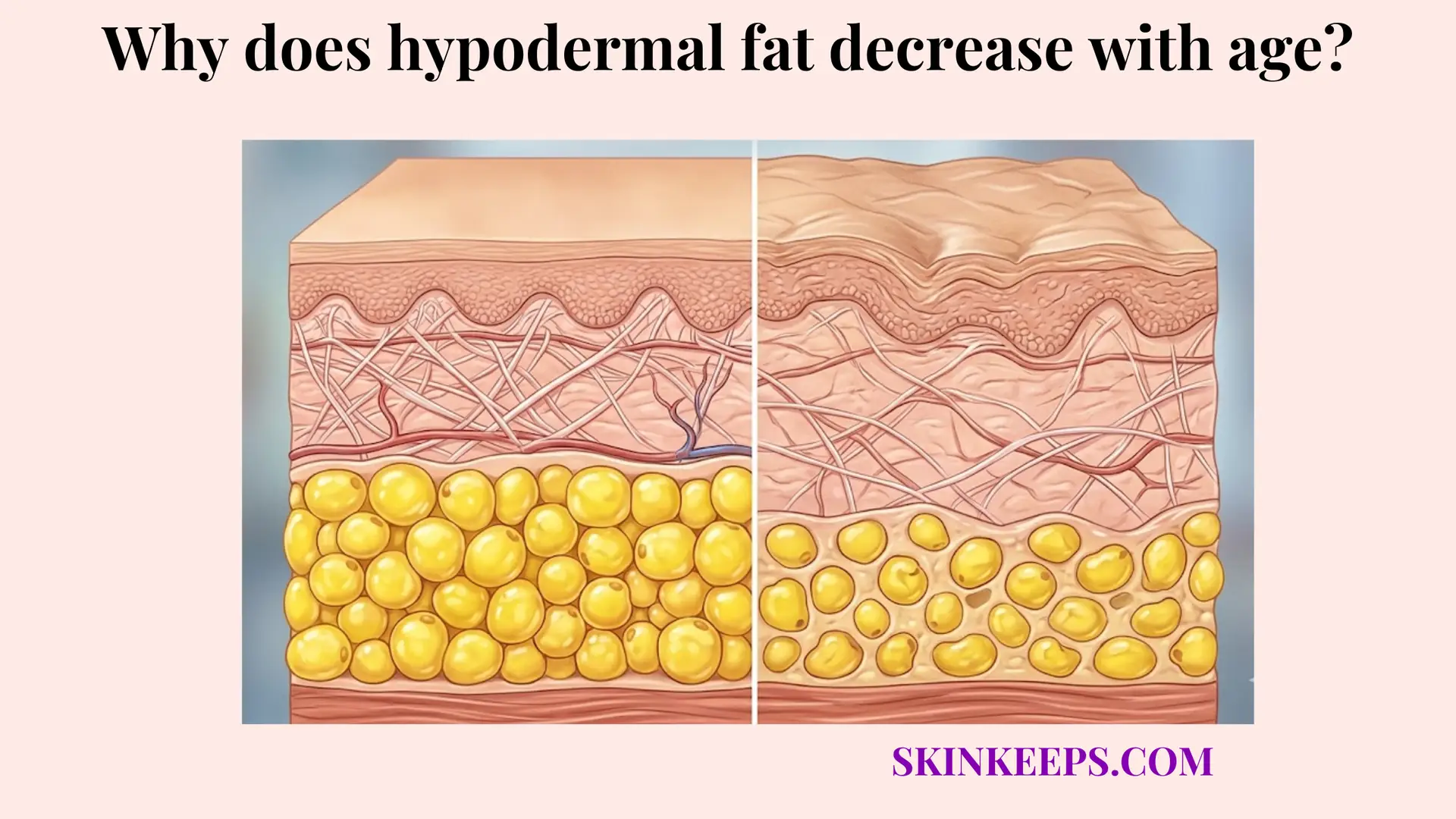
Does aging reduce fat-cell number or just fat-cell size?
Aging ultimately reduces the actual fat-cell number in the hypodermis. While metabolic shifts can also cause cells to shrink in size, the hallmark of structural tissue aging is the failure of the adipogenic renewal process, leading to a net loss of living adipocyte units.
Why is age-related adipocyte loss harder to reverse than ordinary weight loss?
Age-related adipocyte loss is harder to reverse because it involves stem cell exhaustion, progenitor dysfunction, and inflammatory senescence. Ordinary weight loss simply empties the lipid droplets within healthy cells, whereas aging systematically dismantles the cellular population and its capacity to regenerate.
Can dieting regrow lost adipocytes?
No. Dieting or increasing caloric intake can cause existing, living adipocytes to undergo hypertrophy (expand in size) to store more lipids, but it cannot biologically trigger the creation of a new healthy adipocyte population once the structural progenitor cells have been lost to aging.
Does fat grafting actually add new cells?
Yes. Autologous fat grafting physically harvests living adipose tissue, including mature adipocytes and adipose-derived stem cells, from one area of the body and transplants them into the depleted hypodermis, directly adding living cellular units to the tissue.
Do fillers replace lost adipocytes?
No. Dermal and subcutaneous fillers, such as hyaluronic acid, are inert, space-occupying gels. They effectively simulate lost volume and restore visual contours, but they do not contain living cells and cannot replace the missing native adipocyte population.
Why does aging make hollowing look worse even at the same body weight?
Even if total body weight remains stable, aging causes a localized decline in adipocyte number within highly compartmentalized regions like the midface. This specific cellular depletion, combined with matrix degradation, creates stark hollowing that systemic weight cannot mask.
What daily habits help preserve the remaining adipocyte population?
Preserving the remaining adipocyte population requires stabilizing metabolic health, reducing exposure to chronic oxidative stress like UV radiation and smoking, and avoiding rapid, repeated weight-cycling that mechanically damages the supportive adipose niche.
Conclusion
Adipocyte numbers in the hypodermis definitively decline over time due to a profound breakdown in cellular turnover, where the natural rate of apoptosis outpaces the aging body’s ability to generate new fat cells. Protecting the remaining cellular population requires rigorous defense against avoidable metabolic and oxidative stressors that accelerate stem-cell exhaustion. Ultimately, distinguishing true cellular deletion from simple fat shrinkage is essential for selecting appropriate clinical interventions, recognizing that true living-tissue restoration vastly differs from mere volume simulation.