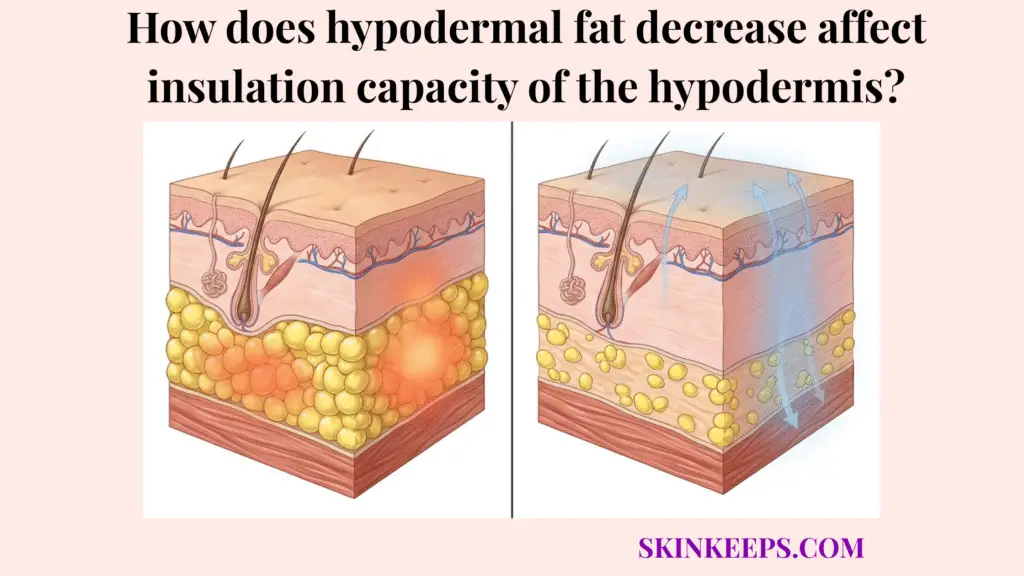
Hypodermal fat decrease affects the insulation capacity of the hypodermis by thinning a low-conductivity biologic barrier that normally slows heat escape from the body. Serving as a crucial layer between the warm internal core and the external environment, the hypodermis fundamentally operates on principles of thermodynamics. By trapping ambient warmth and resisting inward cold transfer, this deep subcutaneous layer acts as the body’s primary passive defense mechanism, shielding vital structures from environmental temperature fluctuations.
When biological aging structurally degrades this lipid-rich barrier, the immediate consequence is a profound loss of passive heat retention. As the adipose envelope thins, the internal metabolic heat generated by the body encounters far less physical resistance, escaping into the colder ambient air at an accelerated rate. Deprived of this crucial hypodermal thermal insulation, the aging body is forced into a state of reactive vulnerability, becoming heavily dependent on energy-costly active compensations—such as restricted blood flow and muscle shivering—just to maintain basic core homeostasis.
What is the baseline thermal insulation capacity of a healthy hypodermis?
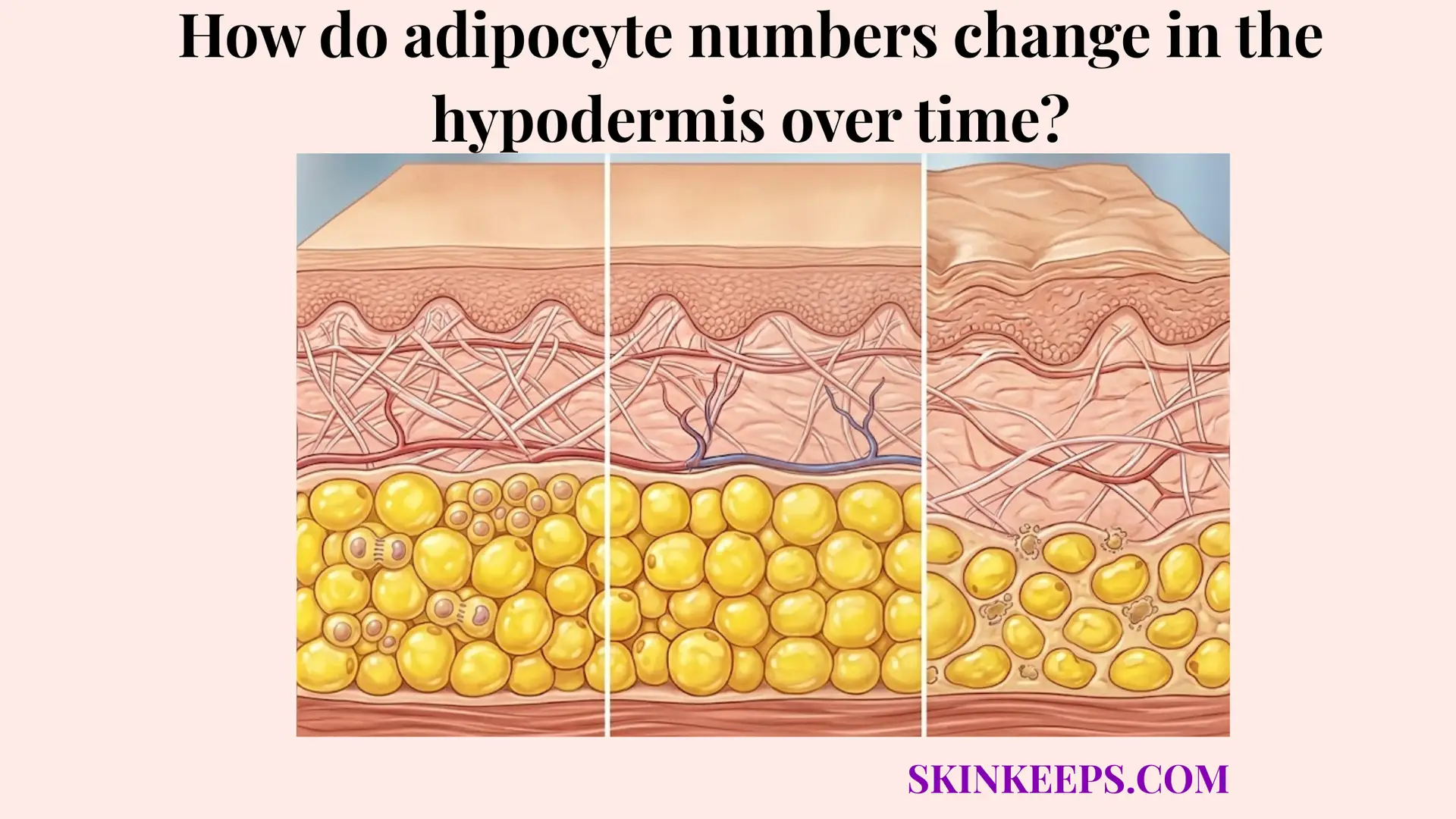
A healthy hypodermis has meaningful thermal insulation capacity because white adipose tissue conducts heat poorly and helps create a thick subcutaneous barrier between the warm core and the colder environment. The hypodermis is the deep subcutaneous layer beneath the dermis containing dense collections of white adipose tissue (WAT)—the body’s main energy-storing fat. Because the biochemical composition of this lipid-rich tissue resists the rapid transfer of kinetic thermal energy, it exhibits an exceptionally low thermal conductivity, meaning it functions exceptionally well as a biological insulation layer.
In the realm of thermophysiology, the effectiveness of an insulating layer is intrinsically tied to its physical thickness. A thick, continuous hypodermal barrier ensures that internal body heat must traverse a significant distance against high thermal resistance before it can reach the surface of the skin and radiate away. The thermal insulation capacity of the hypodermis therefore relies on maintaining adequate subcutaneous depth to continuously blunt the outward flow of vital core warmth.
By possessing the low thermal conductivity of fat, the healthy hypodermis perfectly supports resting systemic homeostasis. While this subcutaneous fat does not actively generate the majority of body heat, it is indispensable for passively preserving the warmth that muscles and organs laboriously produce. Without this robust structural boundary, the body’s baseline energy expenditure would have to rise dramatically just to counteract the constant, unmitigated heat loss to the ambient environment. Subcutaneous fat has been measured at approximately 0.23 W/m·K in thermal-property studies, making it one of the lowest-conductivity human tissues commonly measured, functioning essentially as physiological fiberglass (El-Brawany et al., 2009) [PubMed].
| Tissue type | Thermal behavior | Practical meaning |
|---|---|---|
| Subcutaneous fat | Low conductivity | Better biologic insulation |
| Muscle | Higher conductivity | Heat moves more readily |
| Thin hypodermis | Reduced barrier effect | Faster outward heat loss |
Why does age-related hypodermal fat loss compromise the skin’s ability to retain heat?
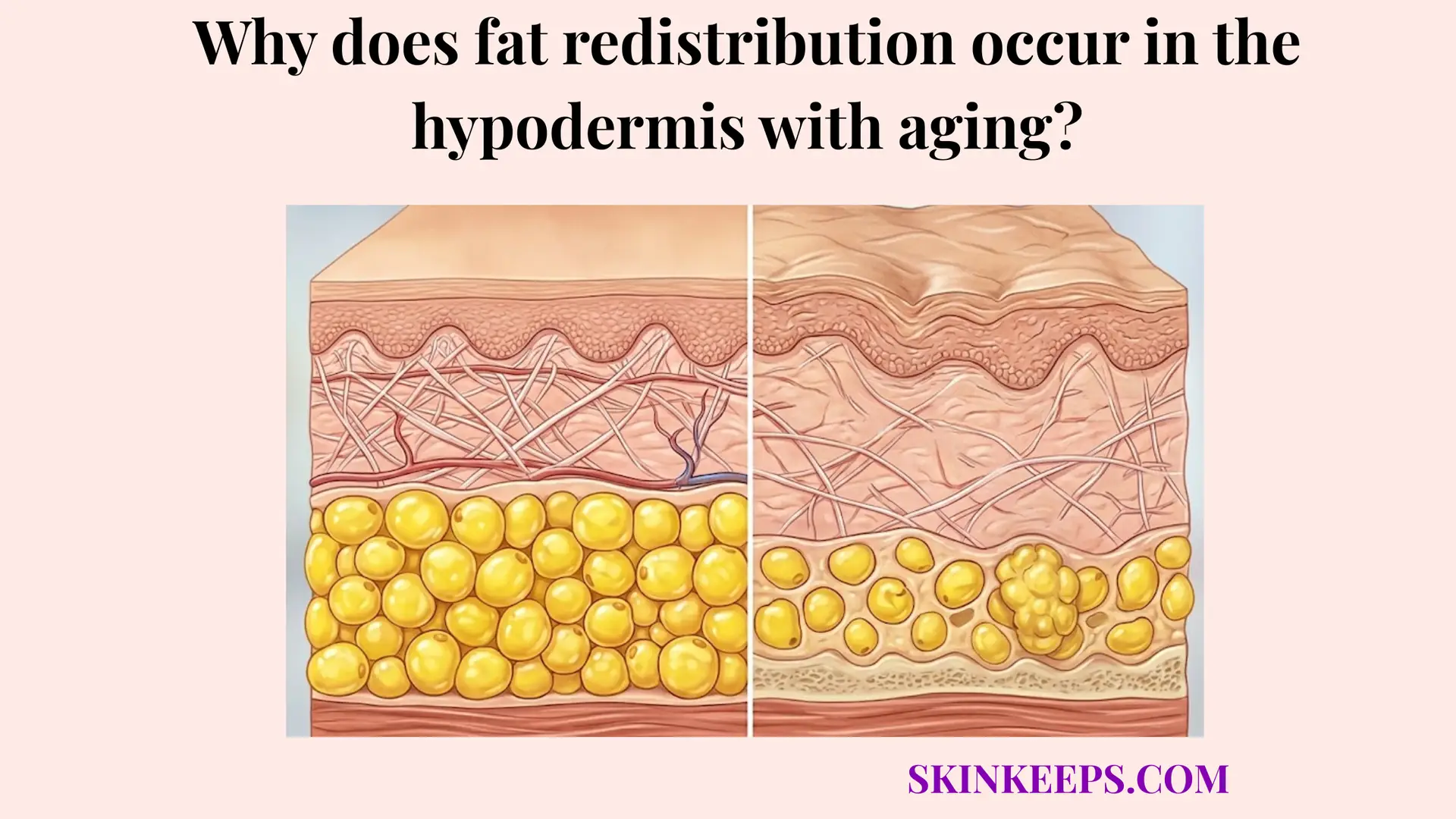
Age-related hypodermal fat loss compromises the skin’s ability to retain heat because the insulating barrier becomes thinner, cutaneous heat transfer becomes easier, and the age-related decline in brown-fat activity reduces one additional source of cold defense. The most direct mechanism of thermal failure is entirely structural. When biological aging permanently shrinks the lipid stores within the deep tissue, the physical distance separating the body’s warm internal core from the unforgiving ambient environment is severely reduced, plummeting the overall thermal insulation capacity of the hypodermis.
Furthermore, this thinning directly exposes the delicate vascular networks to environmental temperature drops. Cutaneous blood flow—blood circulating through the skin vessels—plays a central role in skin blood flow in heat transfer, functioning as the primary route for moving heat from the core to the surface. When the overlying insulating fat is stripped away, these superficial blood vessels lose their biological buffer, allowing the heat carried by the blood to radiate outward into the cold air with alarming efficiency, prompting harsh reactive vasoconstriction.
Finally, maintaining warmth involves more than just passive insulation; it involves active heat generation. Brown adipose tissue (BAT) is a specialized, metabolically active thermogenic fat that generates heat directly through non-shivering thermogenesis. Unfortunately, the brown fat decline with age is a well-documented phenomenon. As older adults lose both the white fat that traps heat and the brown fat that generates it, their systemic defense against cold environments becomes fundamentally compromised on multiple fronts. Thermoregulation references describe skin blood flow as the most effective mechanism for transferring heat from the core to the environment, and human BAT studies consistently report that active BAT significantly declines with advancing age.
How does diminished subcutaneous lipid volume alter your overall systemic thermoregulation?
Diminished subcutaneous lipid volume alters systemic thermoregulation by reducing passive insulation, forcing heavier reliance on active heat production such as shivering, and increasing cold vulnerability in older adults whose overall thermoregulatory reserve is already weaker. When the body’s primary passive defense—the thermal resistance of the fat layer—fails, thermodynamic laws dictate that the core temperature will drop unless active measures are employed. To compensate for this rapid outward heat leakage, the nervous system must drastically upregulate active heat generation.
This biological necessity drives shivering thermogenesis, a high-energy process where repeated, involuntary muscle contractions generate heat to replace what the diminished subcutaneous lipid volume has allowed to escape. Because aging naturally reduces muscle mass alongside fat mass, older adults and cold vulnerability are inextricably linked. Their bodies must work significantly harder to produce warmth, yet they possess fewer metabolic resources to do so, rendering their entire thermoregulatory homeostasis brittle and prone to failure.
Systemically, this translates into severe extremity cooling and profound cold intolerance. The body detects the unmitigated heat loss across the thinned trunk and immediately triggers harsh vasoconstriction in the hands and feet to fiercely protect the core organs. Because the extremities possess naturally lower fat reserves to begin with, this vascular shutdown rapidly turns fingers and toes painfully cold, proving that depleted insulation profoundly dictates where warm blood is allowed to flow. Clinical thermoregulation references define mild hypothermia as a core temperature dropping below 35°C (95°F), specifically identifying older adults as a higher-risk demographic because aging aggressively impairs both heat retention and generation (StatPearls, 2023) [NCBI].
| State | Passive insulation | Active compensation | Clinical implication |
|---|---|---|---|
| Younger / thicker hypodermis | Stronger | Lower dependence | Better cold tolerance |
| Older / thinner hypodermis | Weaker | Greater dependence on shivering and vascular control | More cold sensitivity |
Which metabolic factors actively accelerate the thermal decline of the hypodermis?
The metabolic factors that accelerate the thermal decline of the hypodermis are the ones that either reduce remaining subcutaneous protection or worsen circulation-based heat delivery, especially smoking-related vascular dysfunction, cardiovascular decline, and extreme caloric depletion. Thermal homeostasis requires both an intact physical barrier and efficient fluid dynamics. When chronic smoking introduces heavy toxic oxidative stress into the system, it provokes severe, unnatural vasoconstriction, damaging the microcapillaries that feed the skin and amplifying peripheral heat loss.
Similarly, broad cardiovascular decline drastically undermines the body’s ability to maintain warmth. The hypodermis relies entirely on a steady supply of warm core blood to maintain localized tissue temperatures. When age-related heart failure or arterial stiffening reduces warm-blood delivery to the peripheral tissues, the remaining fat layer becomes effectively useless. An insulating envelope cannot perform its role if the biological furnace fails to deliver heat to the extremities in the first place.
Finally, extreme caloric depletion aggressively accelerates this thermal decline of the hypodermis. While controlled diets manage body weight, severe or repeated underfeeding in older adults cannibalizes the vital protective white adipose reserves that are desperately needed to slow heat loss. Stripping away the body’s final layers of biological insulation severely heightens the risk of systemic temperature drops when confronted with cold ambient environments. Smoking is strongly linked to peripheral vascular dysfunction, heavily amplifying vasoconstrictive issues, while systemic hypothermia risk rises exponentially when structural heat retention and vascular heat delivery simultaneously fail.
What are the biggest myths to avoid when trying to fix depleted hypodermal thermal barriers?
The biggest myths to avoid are the idea that simply gaining weight will rebuild youthful insulation in the right places, and the idea that hot showers somehow train the skin to retain heat better, when both assumptions ignore how aging changes depot distribution and skin-barrier physiology. A dangerous misconception exists that combating age-related thermal loss is as simple as eating more calories. However, biological aging induces central redistribution; excess calories in older adults preferentially accumulate as visceral fat around internal organs, completely failing to rebuild the depleted hypodermal thermal barriers in the limbs and periphery.
Another prevalent error is the hot shower myth. Many individuals suffering from persistent chill attempt to force heat into their bodies through excessively hot bathing. Rather than rebuilding true deep-tissue insulation, scalding water aggressively strips away the epidermis’s vital surface lipid barrier. This hot-water barrier damage vastly accelerates transepidermal water loss (TEWL) and surface dryness, which compromises skin integrity and paradoxically makes the body more vulnerable to environmental stress.
Ultimately, restoring thermal comfort in an aging physiology relies on implementing scientifically valid compensation strategies rather than chasing simplistic, non-biological fixes. Gaining weight in the wrong depots offers no meaningful peripheral warmth, and destroying the superficial skin barrier with heat only worsens cutaneous discomfort, proving that realistic thermoregulation requires understanding the physics of heat loss. Water-temperature research demonstrates that direct hot-water exposure fundamentally damages epidermal barrier function, increasing transepidermal water loss (TEWL) and erythema, proving it harms surface integrity rather than restoring deep fat insulation.
How can you practically manage cold intolerance caused by subcutaneous thermal depletion?
Cold intolerance caused by subcutaneous thermal depletion is managed best by combining external insulation, modest internal heat-generation support, and strategies that reduce unnecessary heat loss, rather than by pretending the lost fat barrier can be fully reversed biologically. Since the physical structure of the biological insulation layer has fundamentally degraded, modern clothing technology must replace it. Utilizing high-performance thermal base layers, such as fine merino wool or engineered synthetics, tightly traps a micro-layer of warm air directly against the skin, serving as exceptional external insulation that functionally compensates for the missing adipose padding.
Nutritional strategy also plays a supportive role through protein-induced thermogenesis. All macronutrients demand energy to digest, but protein boasts a significantly higher diet-induced thermogenesis rate than either carbohydrates or fats. While consuming a protein-rich meal will not magically regrow hypodermal fat, it does modestly raise the body’s internal heat production, supplying a mild but consistent thermogenic metabolic boost that assists the core during cold exposure. The thermic effect of food demands approximately 20–30% of the energy content for protein digestion, versus only 5–10% for carbohydrates and 0–3% for fats, proving protein-rich meals provide superior modest thermogenic support (Moon et al., 2020) [PMC].
Finally, muscle mass compensation for heat loss is paramount. Because skeletal muscle represents one of the body’s largest thermogenic organ systems, preserving or actively building lean muscle tissue dramatically improves baseline heat-generating capacity. When the passive fat barrier fails, possessing dense, metabolically active musculature ensures the body has the robust internal hardware necessary to generate heat and defend its core temperature against invasive environmental cold.
| Intervention type | Mechanism | Replaces fat barrier? | Generates new heat? |
|---|---|---|---|
| Thermal clothing | External insulation | Partly, from outside | No |
| Protein-rich meals | Higher diet-induced thermogenesis | No | Modestly |
| Muscle preservation / training | Improves heat-generating tissue | No | Yes |
| Warm-environment strategy | Reduces heat-loss pressure | No | No |
What is the ultimate checklist for protecting your body against age-related thermal loss?
The ultimate checklist for protecting the body against age-related thermal loss is to accept that cold sensitivity is partly structural, reduce avoidable accelerators of heat loss, and deliberately replace lost biologic insulation with smarter external and internal compensation. The first imperative is acknowledging biological reality: feeling intensely colder with age is frequently not an imagined sensation, but rather the direct thermodynamic result of an irrevocably thinned hypodermal fat layer. Patients must accept this structural depletion to effectively manage it.
Implementing proper compensation means defending the weakest points. Because the physiological barrier is degraded, the body will instinctively restrict blood flow to the fingers and toes to hoard core heat. Therefore, aggressively shielding the extremities with external insulation is paramount. Concurrently, preserving skeletal muscle prevents the internal furnace from shutting down, ensuring the body can actively generate heat when passive lipid retention fails.
Synthesizing this into a sustainable daily routine requires integrating thermal base layers, protecting existing muscle, and optimizing protein intake. This multi-tiered strategy actively compensates for the depleted hypodermal thermal barriers. Gaining unhealthy visceral fat or obliterating the epidermis with scalding showers achieves nothing; only realistic, biology-based thermal compensation ensures enduring physiological comfort against environmental cold. The foundational numbers summarizing this shift are the fat conductivity value of ~0.23 W/m·K (why the physical barrier matters) and the 20–30% thermic-effect range for protein (why internal metabolic heat must modestly compensate) (El-Brawany et al., 2009; Moon et al., 2020) [PubMed].