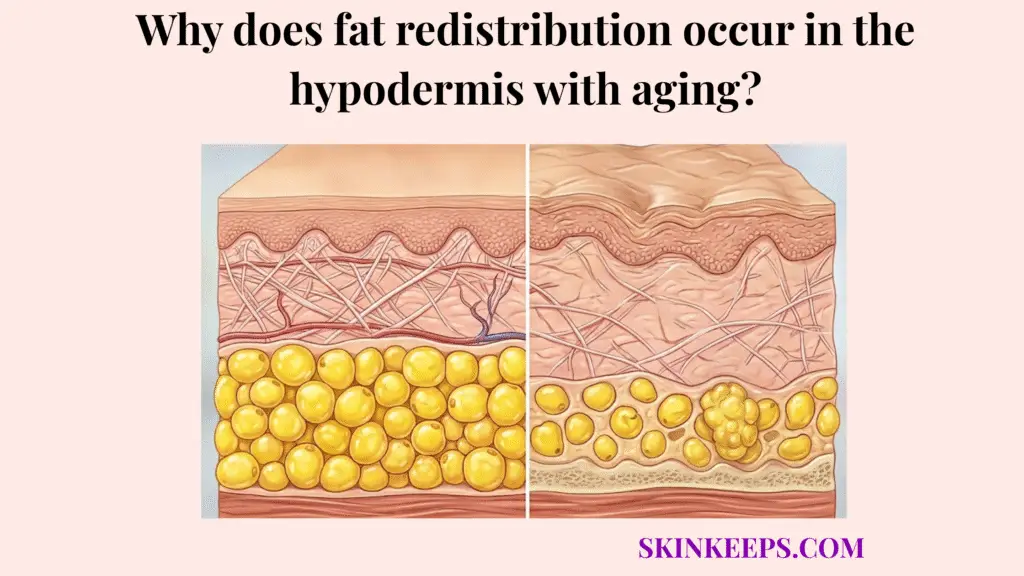
Fat redistribution occurs in the hypodermis with aging because the body gradually shifts away from a younger pattern of more peripheral subcutaneous storage toward a more central pattern, while some facial and limb areas lose soft-tissue fullness and trunk-centered depots become relatively more favored. Rather than a global, uniform expansion or depletion of cellular reserves, biological aging aggressively alters the architectural blueprint of where the body prefers to maintain and deposit its energy. This results in an asymmetrical transformation of the human silhouette that fundamentally differs from simple weight fluctuation driven purely by daily caloric balance.
This shifting behavior explains the visible clinical consequences many patients experience: people can look more hollow in the face, hands, or limbs while simultaneously carrying more fullness centrally, even when their total body weight does not change dramatically. The subcutaneous layer, which once provided a smooth, uniform contour across the appendicular skeleton, progressively surrenders its robust support matrix. At the exact same time, deeper intra-abdominal and central stores become metabolically primed to expand, creating a stark visual contrast that profoundly redefines aging topography.
What exactly is age-related hypodermal fat redistribution?
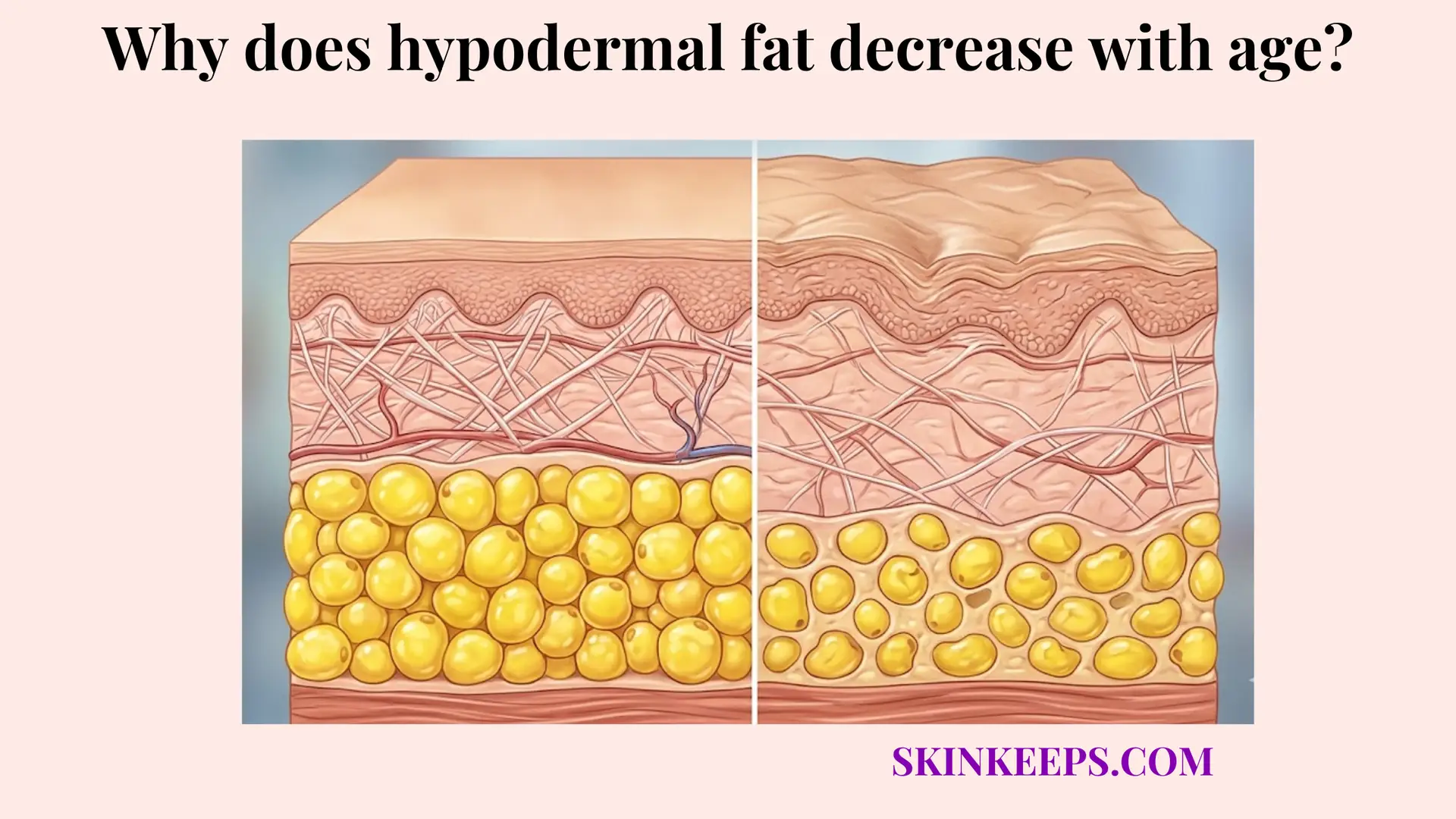
Age-related hypodermal fat redistribution is the physiologic shift in adipose patterning in which some subcutaneous depots lose volume while more central depots, especially visceral and trunk-associated stores, become relatively more prominent with age. Clinicians must properly diagnose this phenomenon as active volume shifting, not simple generalized fat loss or ubiquitous obesity. It is a highly localized, depot-specific cellular phenomenon where the microenvironments supporting distinct fat compartments begin to behave with vastly different metabolic priorities.
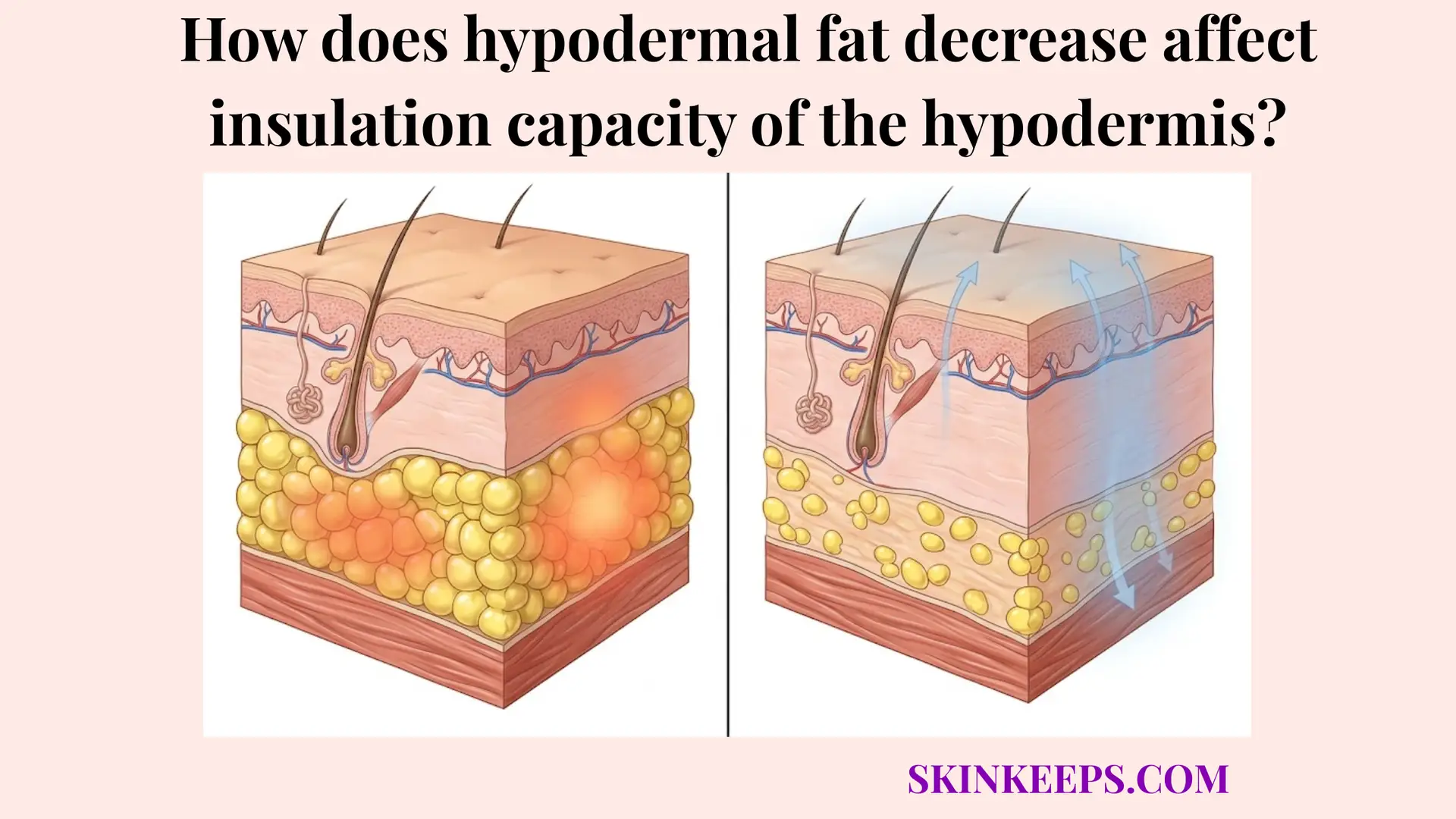
Understanding this transition requires a firm distinction between subcutaneous fat (the soft, pinchable layer residing directly beneath the skin) and visceral fat (the firm, deeper layer packed around internal organs). As aging accelerates, it systematically reduces the vitality and thickness of key peripheral subcutaneous depots, leaving extremities and the face structurally depleted. Conversely, the visceral fat depots experience a surge in relative preservation and active expansion, drastically altering the body’s center of mass.
This opposing physiological trajectory is precisely why someone can notice advanced facial hollowing and prominent abdominal thickening simultaneously, even without a dramatic change on the scale. The volume has not magically relocated; rather, one zone is experiencing structural failure while another is aggressively accumulating mass. Aging reviews report a strong central shift over adulthood, with visceral fat increasing by more than 200% in men and more than 400% in women between the third and seventh decades in one review summary (Hunter et al., 2010) [PMC].
Why does the shifting of subcutaneous fat happen at a biological and hormonal level?
The shifting of subcutaneous fat happens at a biological and hormonal level because aging changes sex-hormone signaling, depot-specific adipose behavior, receptor responsiveness, and insulin sensitivity in ways that favor central storage over peripheral maintenance. Sex-hormone decline serves as the primary catalyst. In healthy youth, abundant estrogen and testosterone tightly regulate fat placement, actively nurturing a peripheral subcutaneous patterning; however, as these hormones decline through processes like menopause and andropause, the biological shield safeguarding peripheral fullness drastically weakens.
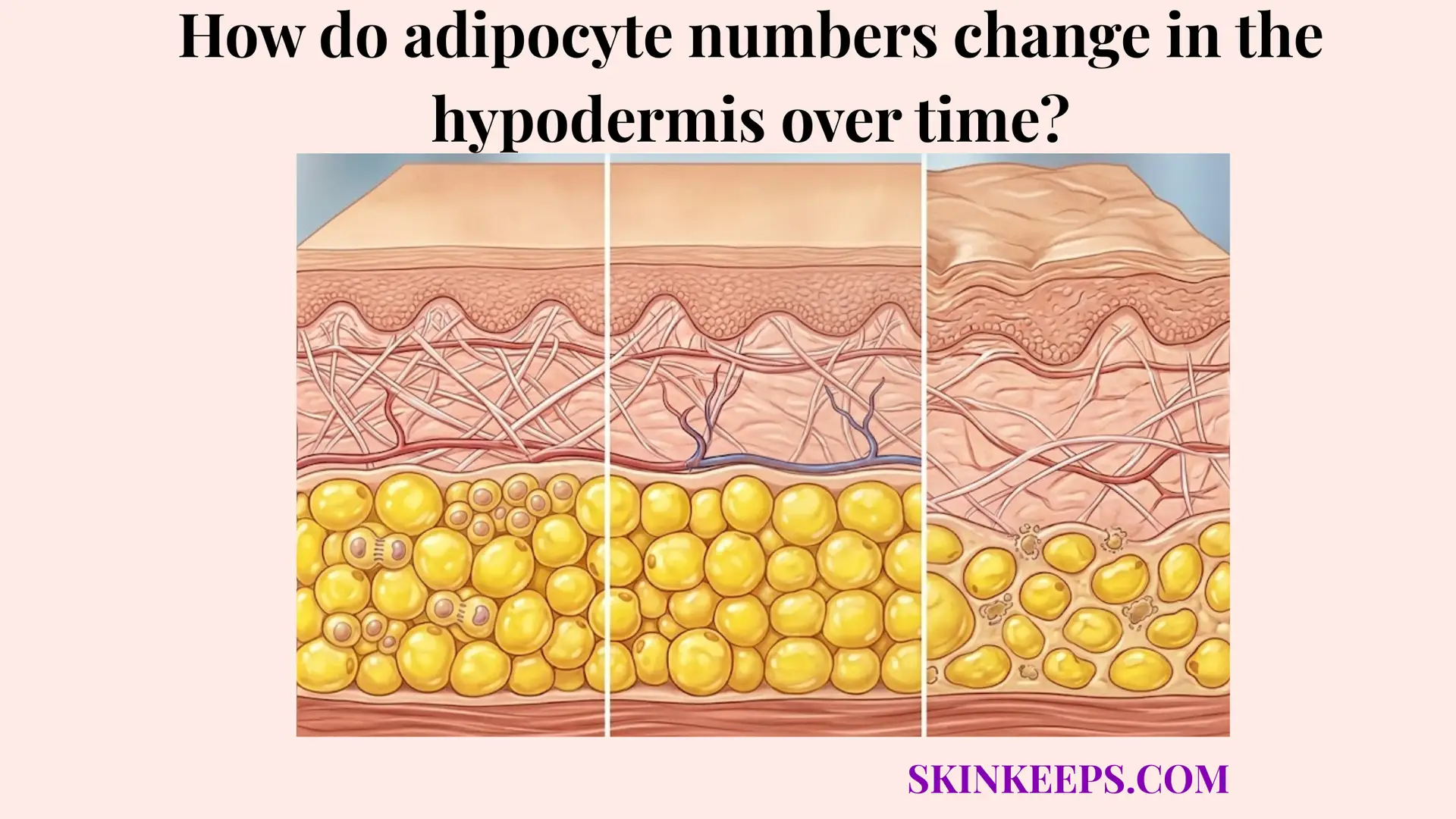
Beneath this hormonal umbrella, depot-specific adipose biology begins to fracture. Adipocytes located in the face or limbs possess different ratios of alpha and beta-adrenergic receptors compared to adipocytes residing in the central trunk. With aging, the receptors governing the trunk depots become exquisitely sensitized to lipid accumulation signals, while the peripheral tissue microenvironments become increasingly prone to senescent degradation and volume deflation. Thus, regional differences in cellular behavior aggressively widen.
Finally, age-related metabolic dysregulation, particularly insulin resistance, firmly anchors this pattern. As peripheral tissues become less responsive to insulin, nutrient partitioning shifts unfavorably. The body becomes less efficient at depositing safe subcutaneous fat and more prone to driving lipids into the central, visceral reservoirs. The downstream pattern of this combined hormonal and metabolic failure is undeniable: visceral fat rises strongly across adulthood, and postmenopausal transition is associated with preferential central fat redistribution (Hunter et al., 2010; Szeliga et al., 2026) [PMC].
| Hormone / driver | Youthful function | Aging dysfunction | Net redistribution effect |
|---|---|---|---|
| Estrogen / sex-steroid support | Helps preserve more peripheral-subcutaneous patterning | Protection weakens with aging and menopause | More central accumulation becomes likely |
| Depot-specific adipose biology | Peripheral depots remain more competitive | Regional differences widen | Uneven loss and gain pattern |
| Insulin sensitivity | Supports healthier nutrient partitioning | Resistance increases with age | More trunk-centered deposition |
| Glucocorticoid signaling | Less dominant central-storage pressure | Chronic stress and cortisol burden amplify central tendency | More abdominal/trunk bias |
Which body regions are most affected by aging-induced adipose displacement?
The body regions most affected by aging-induced adipose displacement are usually the face, dorsal hands, and some peripheral limb depots on the loss side, and the central trunk, lower abdomen, and submental region on the gain side. Formulating a clinical map of this displacement is essential for distinguishing actual aging from generic obesity. On the atrophy side of the spectrum, the highly compartmentalized fat pads of the cheeks and temples systematically thin out, leaving sharp, skeletal facial contours. Concurrently, the back of the hands and selected peripheral limb zones lose their critical subcutaneous padding, unnervingly exposing underlying veins and tendinous structures.
On the opposing hypertrophy side, the displaced energy burden seeks the path of least biological resistance, which aging hormones dictate is the body’s core. The central trunk and lower abdomen experience profound relative gain, swelling with stubborn adiposity that fiercely resists dietary restriction. Additionally, the submental region—the localized area immediately under the chin—frequently accumulates isolated fullness, creating a heavier, less defined jawline even when the upper facial structures remain hollow.
While the exact manifestation is deeply individualized by genetics and lifestyle, the broad direction of this trajectory remains clinically recognizable across the population: peripheral hollowing firmly coupled with intense centralization. To quantify the sheer magnitude of this structural evolution, visceral fat may rise by more than 200% in men and more than 400% in women between the third and seventh decades, robustly supporting the reality of massive central fat redirection (Hunter et al., 2010) [PMC].
| Body zone | Biological action | Visible effect |
|---|---|---|
| Cheeks / temples | Relative loss / atrophy | Hollowing, flatter contours |
| Dorsal hands | Relative loss / thinning | More visible veins, tendons, joints |
| Peripheral limbs | Relative subcutaneous depletion | Leaner or less padded look |
| Central trunk | Relative gain | Thicker waist or central fullness |
| Lower abdomen | Relative gain | More persistent central storage |
| Submental region | Localized gain | Fuller under-chin contour |
Which metabolic stressors actively accelerate this hormonal fat relocation?
The metabolic stressors that actively accelerate this hormonal fat relocation are the ones that worsen cortisol exposure, insulin resistance, and circadian disruption, because those pressures favor more central fat storage and weaken the biology that protects peripheral fullness. Chronic psychological and physiological stress acts as a master switch. Cortisol biology is not merely a cosmetic myth; specific central fat depots maintain elevated densities of glucocorticoid receptors that respond profoundly to sustained stress hormones, actively sequestering lipids around the abdomen while starving peripheral fat pads.
Simultaneously, chronic sleep deprivation and circadian misalignment inflict immense metabolic damage. Disrupting natural sleep cycles severely impairs systemic insulin sensitivity, forcing the body to secrete higher volumes of insulin to manage blood glucose. This hyperinsulinemic state creates a heavily biased physiological environment that explicitly favors visceral and central-trunk adiposity, systematically accelerating the volume shifting away from healthy subcutaneous storage.
In practical synthesis, a high-stress lifestyle combined with poor rest does not literally teleport fat cells from the face directly into the belly overnight. Rather, these stressors relentlessly strengthen the central-storage signaling pattern over years, cementing the aged, hollowed-periphery look. Emphasizing this impact, even a single night of partial sleep deprivation has been shown to induce measurable insulin resistance in healthy subjects, explaining precisely why chronic sleep disruption can aggressively bias long-term fat distribution (Mesarwi et al., 2013) [PMC].
Which clinical procedures effectively correct age-altered fat placement?
The clinical procedures that most effectively correct age-altered fat placement are those that either reposition living fat, remove localized excess, or camouflage loss, depending on whether the main problem is central accumulation, peripheral hollowing, or both. Proper medical aesthetic correction respects the specific directional nature of the volume shift. Autologous fat grafting serves as the ultimate “remove and replace” strategy, offering a biologically identical solution by gently harvesting misplaced excess from central trunk depots and surgically restoring that living volume to hollowed facial or hand tissues.
Conversely, targeted fat reduction relies on entirely different mechanisms to manage stubborn hypertrophic zones. Techniques like precision liposuction physically evacuate structural excess where central gain heavily dominates. In more localized anatomical contexts, such as the submental region under the chin, injectable cytolytic agents like deoxycholic acid can be meticulously administered to chemically dismantle the expanding adipose membrane, permanently destroying those specific fat cells.
Finally, when direct living-tissue transfer is deemed unnecessary or inappropriate, structural camouflage remains a primary tool. Advanced dermal fillers expertly simulate volume, providing an optical lift to severely hollowed zones, though it is paramount to understand they do not literally relocate misplaced fat. Highlighting localized destruction logic, deoxycholic acid causes irreversible adipocyte destruction in the treated area and is used specifically for submental fat reduction, definitively altering that specific accumulation pattern (Inocêncio et al., 2023; Mayo Clinic) [PMC].
| Treatment type | Area targeted | Mechanism | Best for |
|---|---|---|---|
| Autologous fat grafting | Hollow face, hands, selected soft-tissue deficits | Add / replace living volume | Loss in one area with gain elsewhere |
| Liposuction | Localized trunk or under-chin excess | Remove | Structural excess where central gain dominates |
| Deoxycholic acid | Submental fat | Remove | Small, localized under-chin accumulation |
| Dermal filler | Hollow areas | Camouflage / add volume effect | When direct transfer is not needed or not chosen |
What are the biggest myths to avoid when trying to reverse hypodermal volume shifting?
The biggest myths to avoid are the belief that spot reduction can fix hormone-driven central gain and the belief that severe dieting will restore youthful distribution, when in reality both strategies can worsen the contrast between central accumulation and peripheral hollowing. The fitness and wellness industries heavily perpetuate the spot-reduction fallacy. Performing endless localized abdominal exercises strictly strengthens underlying musculature; it absolutely cannot command the overlying hormone-driven central fat depots to selectively empty themselves while ignoring the rest of the body.
Equally dangerous is the extreme-calorie-deficit myth. Because age-related redistribution has already rendered peripheral subcutaneous tissue extremely fragile, embarking on an aggressive, starvation-style diet is disastrous for aesthetic contours. Severe caloric restriction aggressively strips whatever remaining fullness is left in the face, temples, and hands long before it meaningfully addresses stubborn visceral belly fat, leaving the patient looking prematurely skeletal rather than youthful.
Ultimately, pursuing the right restorative approach demands a nuanced assessment of whether the patient’s primary concern is where fat has accumulated, where it has been lost, or a combination of both. Misdiagnosing the condition dictates poor intervention. Confirming the risk of improper strategy, massive weight loss is strongly associated with severe facial fat devolumization and profound structural laxity, explaining exactly why aggressive dieting often makes redistribution look visibly worse in the face (Jafar et al., 2024) [PMC].
What is the ultimate daily checklist for managing natural subcutaneous fat transfer?
The ultimate daily checklist for managing natural subcutaneous fat transfer is to reduce the biologic forces that push fat centrally, while protecting peripheral fullness from unnecessary depletion and treating true redistribution with realistic, region-specific decisions. Biologically managing this shift requires stabilizing insulin and cortisol pressure through stringent sleep regularity, proactive stress control, and highly sustainable metabolic eating habits. Preventing the biochemical triggers that command central storage is the only method to decelerate the process internally.
Patients must heed the critical warning that “eat less and move more” is an insufficient protocol for addressing targeted architectural atrophy. Severe dieting strips essential peripheral and facial fullness significantly faster than it remedies abdominal accumulation. Therefore, the daily management goal is strictly not to indiscriminately starve the body, but rather to foster an environment where remaining subcutaneous padding is preserved against mechanical and metabolic degradation.
If true aesthetic correction is required, patients must navigate their interventions based on precise hormone-aware management, prioritizing region-specific reduction and region-specific restoration over one crude, global fat-loss effort. Emphasizing the metabolic baseline, inadequate sleep is tightly associated with impaired insulin sensitivity and heavily visceral-adiposity–favoring physiology, proving that daily systemic habits directly script your structural fat map (Mesarwi et al., 2013; Hunter et al., 2010) [PMC].
Fat Redistribution Management Checklist
Clinical Key Takeaway
Age-related hypodermal fat redistribution is an independent endocrine event, distinctly separate from simple caloric weight gain. It mandates a dual-focused aesthetic strategy: halting the metabolic accelerators of central adiposity while employing targeted, region-specific procedures (like fat grafting or dermal fillers) to address the inevitable peripheral hollowing.
Frequently Asked Questions About Fat Redistribution
Is age-related fat redistribution just generic weight gain?
No. Generic weight gain typically involves a proportional expansion of fat cells across the body. Age-related fat redistribution is a pattern shift where peripheral areas like the face and hands lose subcutaneous fat, while central zones like the trunk and visceral compartments actively accumulate it.
Does abdominal fat literally migrate from the face?
Fat does not mechanically migrate or travel from the face to the abdomen. Instead, the local cellular environments change. The biological support for facial subcutaneous fat weakens leading to atrophy, while the central depots become hormonally primed to store more energy, leading to hypertrophy.
How does cortisol affect fat shifting?
Chronically high cortisol levels increase glucocorticoid signaling, which strongly favors lipid accumulation in central and visceral depots. This chronic stress response actively biases the body to store energy around the trunk rather than in the peripheral subcutaneous layers.
Can dieting reverse facial hollowing caused by redistribution?
No. Severe or aggressive dieting often worsens facial hollowing because a systemic caloric deficit will continue to shrink the remaining, already-fragile peripheral fat pads. It cannot selectively target central fat while ignoring peripheral fat.
Do dermal fillers replace misplaced fat?
Dermal fillers do not replace actual fat cells. They are space-occupying gels used as structural camouflage to lift and smooth hollowed zones. Only autologous fat grafting physically transfers living adipose tissue to depleted peripheral areas.
Can targeted exercise remove hormonal belly fat?
Spot reduction through exercise is a physiological myth. While systemic exercise improves overall metabolic health and insulin sensitivity—which can indirectly help regulate central fat—doing localized abdominal exercises cannot selectively burn off hormone-driven central adiposity.