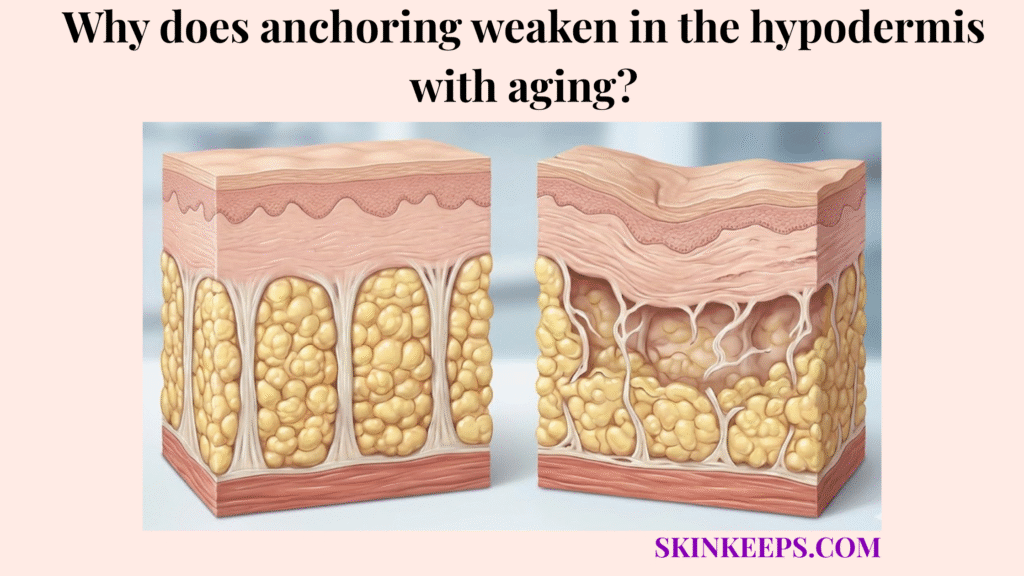
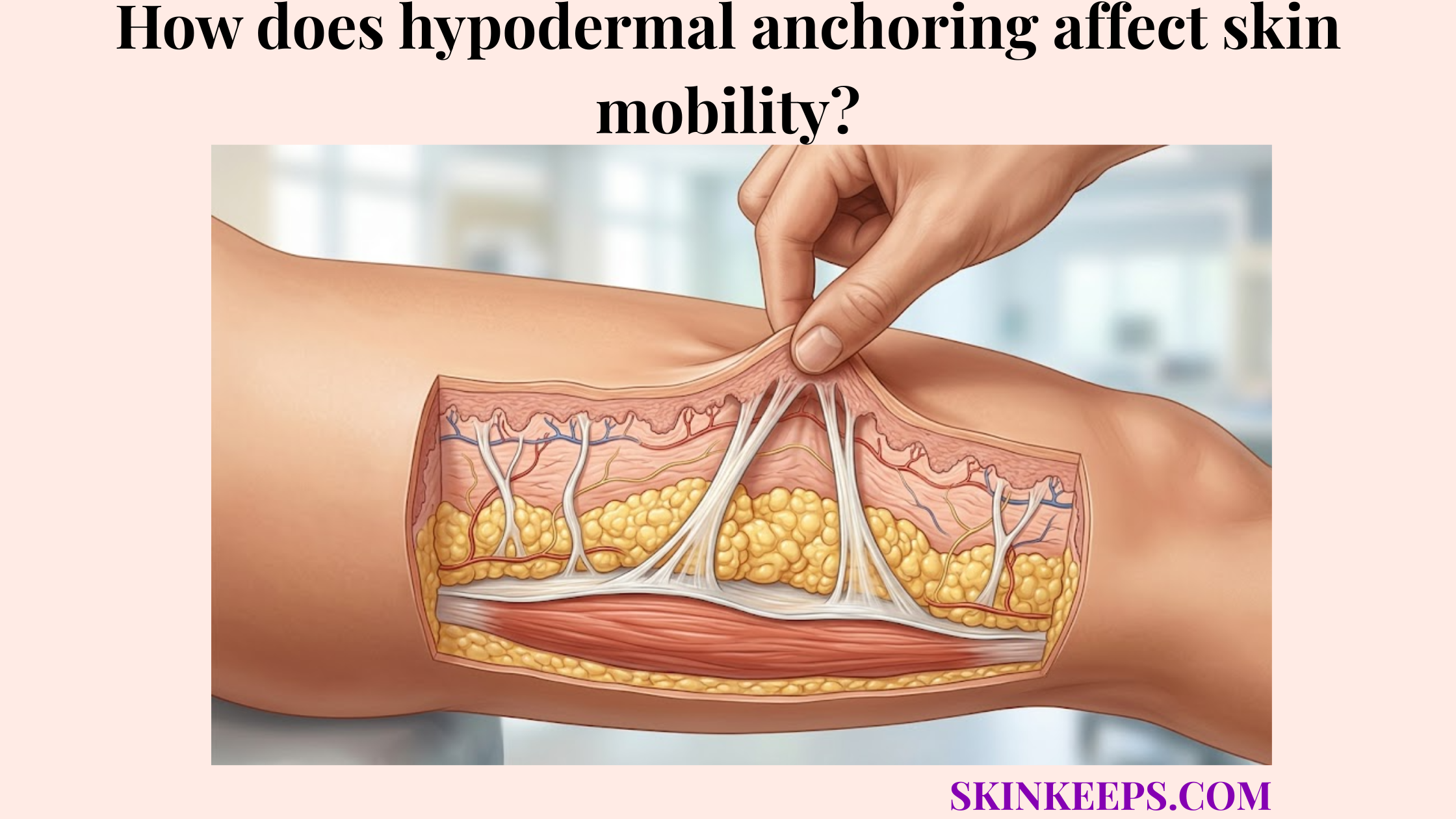
Hypodermal anchoring weakens with aging because collagen-rich fibrous septae, elastin fibers, fibroblast activity, and dermal-fascial support gradually lose strength, recoil, and repair capacity. This age-related weakening transforms a once-resilient subcutaneous tethering system into a more fragile, less supportive network.
This guide covers the youthful baseline of deep connective anchors and explains how menopause, MMP activity, glycation, and UV exposure accelerate tissue changes. We also separate anti-aging myths from reality, evaluating regenerative treatments and daily preservation habits that support structural longevity.
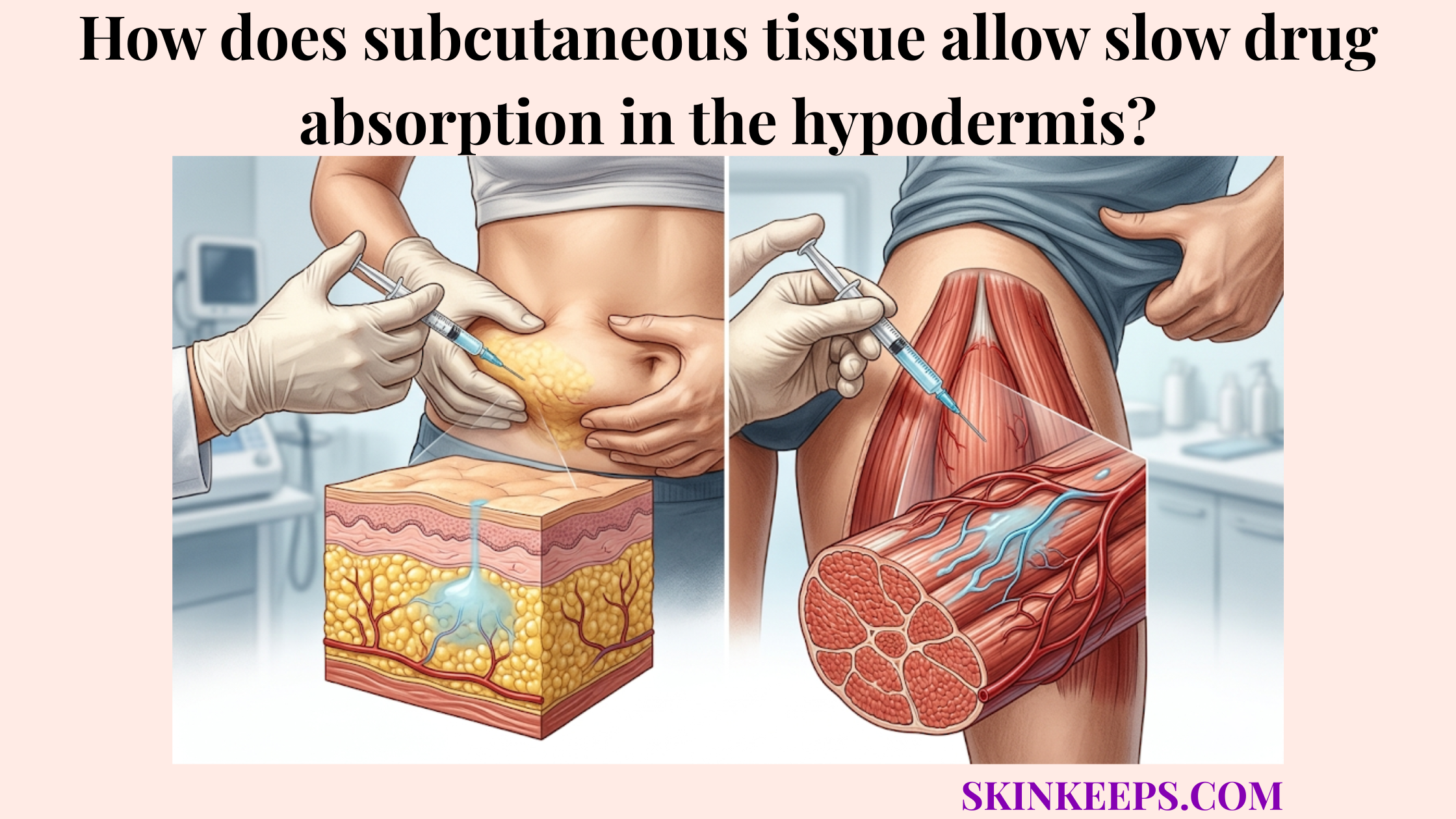
What happens to the structural integrity of subcutaneous tethering as we age?
The structural integrity of subcutaneous tethering changes with age as collagen fibers become less organized, elastin recoil declines, fibroblast activity shifts, and the anchoring network loses some tensile strength. The hypodermis supports adipose storage, insulation, and structural anchoring by relying on this network of fascial connective tissue.
As this deep support system slowly degrades, the overall architecture begins to yield under normal mechanical loads.
How does collagen fragmentation weaken hypodermal anchors?
Collagen fragmentation weakens hypodermal anchors by reducing the organized tensile network that normally helps fibrous septae hold the dermis, fat, and fascia in stable alignment. In youthful tissue, thick bands of type I collagen provide robust tensile support.
With aging, fibroblast senescence means these cells produce less new collagen and struggle to repair micro-tears efficiently. This gradual fragmentation reduces tensile support, contributing to visible tissue laxity and reduced firmness.
How does elastin loss reduce anchor recoil?
Elastin depletion reduces anchor recoil because elastin helps connective bands stretch and return toward their resting shape after mechanical load. In a healthy extracellular matrix, elastin fibers allow the anchoring network to bounce back after daily facial movement or gravity-induced stretching.
Early or progressive elastin damage may reduce this snap-back ability over decades of use. This loss of recoil capacity directly contributes to looseness and stretched, sagging tissue.
| Feature | Youthful Anchoring Network | Aging Anchoring Network | Visible / Functional Meaning |
|---|---|---|---|
| Collagen organization | Dense, better organized support fibers | More fragmented or less organized fibers | Reduced tensile support |
| Elastin recoil | Better stretch-and-return behavior | Reduced recoil after repeated loading | More laxity or looseness |
| Fibroblast activity | More efficient matrix maintenance | Slower or altered matrix remodeling | Reduced repair capacity |
| Septal tension | Stronger dermis-fat-fascia connection | Weaker or less resilient tension | Sagging may become more visible |
| Mechanical response | Better force distribution | Less efficient recovery after stress | Tissue may shift or stretch more easily |
Why do hormonal shifts during menopause accelerate degradation of deep connective anchors?
Hormonal shifts during menopause may accelerate degradation of deep connective anchors because lower estrogen signaling is associated with reduced collagen production, thinner skin, and decreased connective tissue resilience. During the transition into perimenopause and menopause, the entire skin collagen context changes as hormonal support drops.
Because collagen density decreases in the dermis with age, this systemic shift can make age-associated tissue laxity much more apparent.
How does estrogen support fibroblast and collagen activity?
Estrogen supports fibroblast and collagen activity by influencing skin thickness, extracellular matrix renewal, hydration, and collagen maintenance across hormonally responsive skin tissues. Estrogen receptors on fibroblasts help sustain efficient collagen synthesis and healthy tissue repair.
As estrogen decline progresses, this biological support diminishes, making the connective tissue less able to maintain its original structural integrity.
How much collagen can decline after menopause?
Collagen can decline more quickly after menopause, which helps explain why skin laxity, dryness, and reduced firmness may become more visible during this life stage. Women’s skin loses about 30% of its collagen during the first 5 years of menopause, followed by about 2% collagen loss each year for the next 20 years. [AAD]
| Life Stage | Estrogen Context | Fibroblast / Collagen Context | Structural Anchor Meaning |
|---|---|---|---|
| Reproductive years | Higher cyclic estrogen | Better collagen-support environment | Stronger tissue resilience |
| Perimenopause | Fluctuating estrogen | Collagen maintenance may become less stable | Early laxity may appear |
| Early menopause | Lower estrogen | Faster skin collagen decline may occur | Reduced firmness becomes more visible |
| Later menopause | Long-term estrogen deficiency | Gradual collagen and elastin changes continue | Laxity and dryness may progress |
| Clinical care | Individual risk profile matters | HRT is medical, not cosmetic-only | Discuss only with a physician |
How do matrix metalloproteinases chemically weaken hypodermal retaining ligaments over time?
Matrix metalloproteinases can chemically weaken hypodermal retaining ligaments over time by degrading extracellular matrix proteins when collagen breakdown exceeds collagen repair. These MMPs are normal remodeling enzymes that help clear out old, damaged tissue.
However, aging often shifts the balance toward excessive enzymatic degradation, slowly diminishing the quality of the anchoring network.
How does the balance between MMPs and TIMPs affect anchor strength?
The balance between MMPs and TIMPs affects anchor strength because MMPs degrade matrix proteins while TIMPs help restrain excessive matrix breakdown. Tissue inhibitors of metalloproteinases (TIMPs) naturally regulate MMP-1 and other remodeling enzymes.
When this MMP-TIMP regulation becomes imbalanced, the enzymes degrade more collagen than the fibroblasts can efficiently replace. This imbalance regulates matrix integrity, leading to a progressively weaker collagen network.
How does inflammaging keep collagen breakdown active?
Inflammaging can keep collagen breakdown active by maintaining low-grade inflammatory signaling that may increase matrix-remodeling enzymes and oxidative stress. This chronic, low-grade inflammation associated with aging creates a harsh environment for deep structural supports.
By sustaining an elevated level of breakdown signals, inflammaging increases matrix remodeling pressure and weakens overall tissue resilience.
Which external lifestyle factors prematurely weaken aging fascial attachments?
External lifestyle factors may prematurely weaken aging fascial attachments by increasing oxidative stress, glycation, UV-related enzyme activity, and chronic inflammatory pressure on collagen-rich support tissues. These extrinsic accelerators push the biological tissue beyond its normal aging trajectory.
Because UV radiation degrades dermal collagen, protecting the skin from these stressors helps maintain a more resilient hypodermal framework.
How do advanced glycation end-products stiffen collagen-rich anchors?
Advanced glycation end-products can stiffen collagen-rich anchors by forming cross-links in long-lived structural proteins, which may reduce flexibility and elastic recovery. This glycation process occurs when excess sugars bind to collagen fibers, leading to glycation-related stiffness.
These AGEs cross-link collagen proteins, turning once-pliable retaining bands into brittle structures that are more susceptible to mechanical failure.
How does UVA-driven photoaging affect deep connective support?
UVA-driven photoaging affects deep connective support by promoting oxidative stress and matrix-degrading enzyme activity that can weaken collagen and elastin quality over time. Deeper-penetrating UV radiation can trigger molecular responses that affect the structural layers holding the skin.
This chronic photoaged connective tissue stress promotes ongoing collagen degradation and visible laxity.
What myths should be avoided when trying to reverse aging of dermal-fascial bonds?
Myths should be avoided when trying to reverse aging of dermal-fascial bonds because deep connective tissue aging cannot be fully corrected by surface creams, collagen drinks, or aggressive self-treatment. The cosmetic industry often markets topical products as capable of rebuilding senescent deep-tissue anchors.
Separating surface support from deep structural repair ensures patients pursue realistic improvement strategies.
Why can’t topical collagen rebuild senescent deep-tissue anchors?
Topical collagen cannot rebuild senescent deep-tissue anchors because collagen applied to the skin surface cannot mechanically reconstruct the retinacula cutis or hypodermal septal network. These products generally hydrate the epidermis and support surface texture, but they do not penetrate to the subcutaneous tissue.
Topical collagen supports the surface but cannot rebuild deep anchors or reverse structural aging.
Why can dietary collagen not guarantee deep anchor repair?
Dietary collagen cannot guarantee deep anchor repair because collagen peptides are digested into amino acids and peptides that the body distributes according to broader metabolic needs. While they may provide nutritional building blocks for collagen synthesis, they do not selectively target loose fibrous septae.
Dietary collagen supplies nutrients but does not function as a targeted reconstructive treatment for sagging skin.
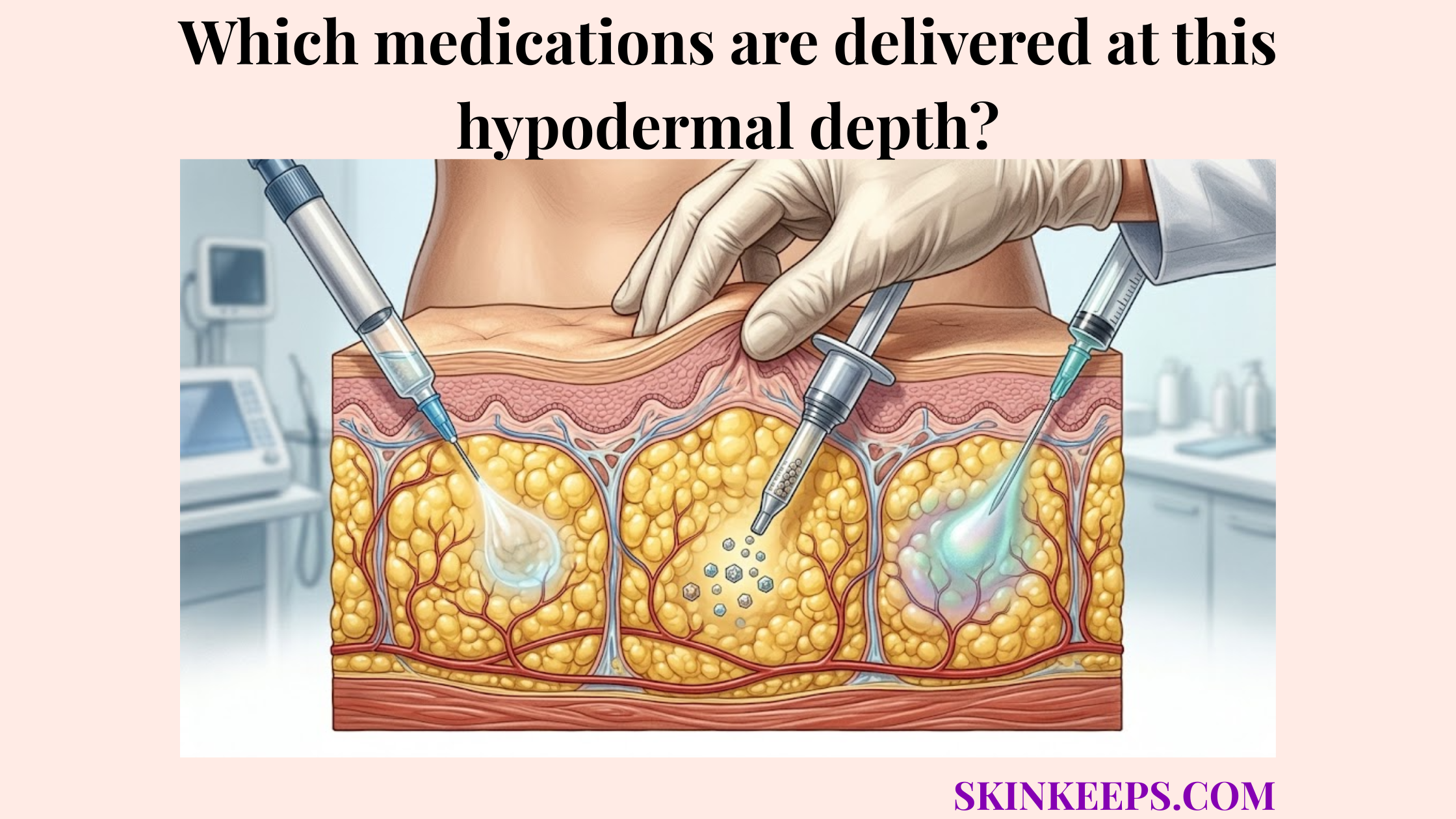
Which regenerative clinical treatments may support a senescent subdermal anchoring network?
Regenerative clinical treatments may support a senescent subdermal anchoring network by stimulating collagen remodeling, improving skin quality, or strengthening selected support layers in clinician-selected patients. Because treatments stimulate collagen production in the dermis and underlying fascia, they offer a biomechanical approach to age-related laxity.
These therapies aim to support the remaining tissue rather than instantly replacing youth.
How do PLLA and CaHA support collagen biostimulation?
PLLA and CaHA support injectable biostimulation by creating a controlled tissue response that may increase collagen remodeling and improve skin quality in selected patients. Poly-L-lactic acid (PLLA) and calcium hydroxylapatite (CaHA) act as biostimulators that encourage local fibroblasts to generate new structural proteins.
These collagen stimulators support collagen remodeling over several months, making them a clinician-guided option for deep connective tissue support.
How does focused thermal remodeling support aging anchors?
Focused thermal remodeling may support aging anchors by using ultrasound or radiofrequency energy to heat selected tissue layers and stimulate collagen remodeling over time. Microfocused ultrasound (MFU-V or HIFU) delivers precise thermal coagulation points into the deeper connective planes.
MFU-V devices can use focal depths of 1.5 mm, 3.0 mm, and 4.5 mm, which supports describing the technology as layer-targeted rather than a surface-only treatment. [JCAD]
| Treatment Type | Main Target | Mechanism on Aging Tissue | Expected Longevity Language |
|---|---|---|---|
| PLLA | Dermal/subdermal collagen remodeling | Gradual collagen biostimulation | Results develop over time; varies by patient |
| CaHA | Skin quality and laxity support | Collagen-stimulating scaffold effect | May improve firmness in selected patients |
| MFU-V / HIFU | Selected dermal/subdermal depths | Focused thermal remodeling | May improve laxity; results vary |
| Radiofrequency | Dermal and subdermal support | Heat-based collagen remodeling | May improve firmness or texture |
| Retinoids | Dermal fibroblast biology | Topical dermal support | Does not rebuild deep anchors |
| HRT | Systemic menopause care | Medical hormone management | Not a cosmetic anchoring treatment; physician-guided only |
What daily checklist helps delay biological weakening of deep structural supports?
A daily checklist helps delay biological weakening of deep structural supports by reducing UV exposure, supporting collagen maintenance, managing inflammation risk, and avoiding habits that add unnecessary mechanical stress. Consistent preservation habits protect the extracellular matrix from accelerated decay.
This structural longevity checklist focuses on safely managing the tissue environment over time.
How can inflammation and endocrine health be managed safely?
Inflammation and endocrine health can be managed safely by treating systemic symptoms through qualified medical care while using lifestyle habits that support general tissue resilience. Clinician-guided menopause care and hormone replacement therapy (HRT) may address systemic hormonal changes, but these should be discussed only with a physician for health symptoms, not solely as cosmetic anchoring treatments.
Medical guidance supports safe endocrine care while reducing chronic inflammatory triggers.
How can nutrition and environmental defense protect anchoring collagen?
Nutrition and environmental defense can protect anchoring collagen by supporting normal extracellular matrix maintenance and reducing avoidable UV, oxidative, and glycation-related stress. A balanced diet provides essential amino acids and vitamin C for proper collagen synthesis without the need for strict diets.
Utilizing broad-spectrum sunscreen protects the dermal envelope, while retinoids may support general skin quality.
Daily Deep Tissue Longevity & Preservation Checklist
FAQs About Aging and Hypodermal Anchoring
What does hypodermal anchoring weakening mean?
Hypodermal anchoring weakening means the collagen-rich septae and connective structures that link the dermis, fat, and fascia lose some strength, recoil, and repair capacity. This can make skin support less firm and age-related laxity more visible.
Does menopause weaken skin support?
Menopause can affect skin support because lower estrogen is associated with reduced collagen production, dryness, thinning, and elasticity changes. Women’s skin loses about 30% of collagen during the first 5 years of menopause. [AAD]
Do MMP enzymes destroy hypodermal anchors?
MMP enzymes do not simply destroy anchors. They are normal matrix-remodeling enzymes, but aging, UV exposure, or inflammation may shift the balance toward more collagen breakdown than repair, which can weaken connective tissue quality over time.
Can sugar make anchoring tissue weaker?
High glycation exposure may contribute to connective tissue stiffness because AGEs can cross-link long-lived proteins such as collagen. This does not mean sugar instantly damages anchors, but metabolic health can influence long-term skin aging biology.
Can topical collagen rebuild weakened anchors?
No. Topical collagen may support hydration or surface texture, but it cannot mechanically rebuild deep retinacula cutis or hypodermal septae. Deep anchoring support depends on extracellular matrix remodeling below the surface.
Can collagen supplements repair hypodermal anchoring?
Collagen supplements may provide amino acid building blocks, but they do not guarantee targeted repair of hypodermal anchoring. The body distributes nutrients according to broad metabolic needs, so supplements should be framed as supportive, not reconstructive.
Which treatments may support aging anchors?
Biostimulators, radiofrequency, HIFU, and MFU-V may support collagen remodeling in selected patients. MFU-V devices can use focal depths of 1.5 mm, 3.0 mm, and 4.5 mm, depending on device protocol. [JCAD]
Conclusion
Hypodermal anchoring weakens with aging because collagen, elastin, fibroblast activity, hormonal support, and extracellular matrix balance gradually change over time. These changes can reduce tissue recoil, firmness, and dermal-fascial support, especially when UV exposure, glycation, inflammation, or rapid tissue stress adds extra burden.
At SkinKeeps, we explain skin aging through evidence-based dermatology and anatomy. Understanding hypodermal anchoring helps readers choose realistic prevention habits and clinician-guided treatments without exaggerated anti-aging promises.