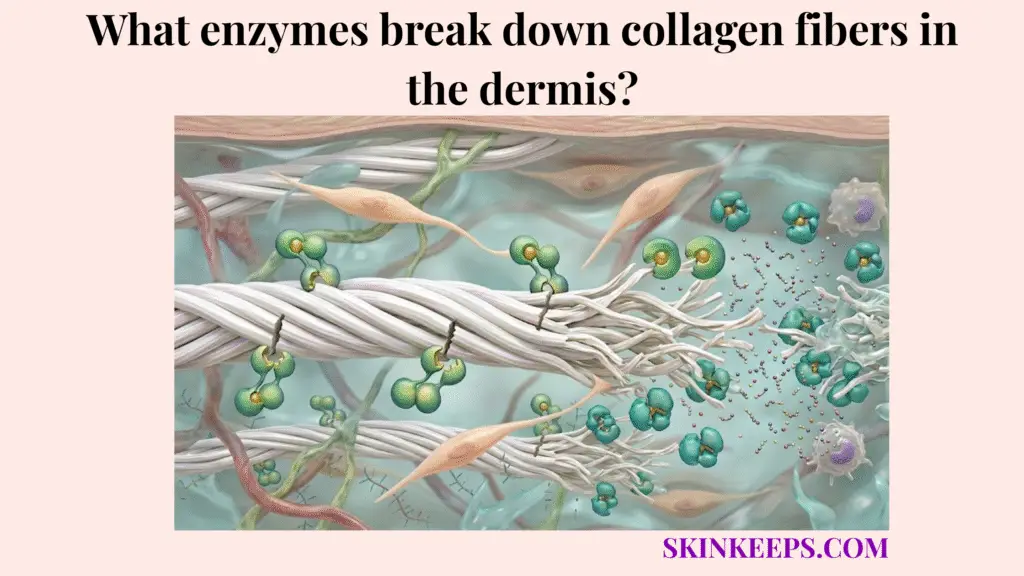
The main enzymes that break down collagen fibers in the dermis are Matrix Metalloproteinases (MMPs), especially MMP-1, which acts as the key collagenase that starts dermal collagen cleavage. Other enzymes such as MMP-3 and MMP-9 help continue the breakdown process after the initial structural cut.
This collagen loss is an active enzymatic destruction process, not only a production slowdown. While aging reduces how fast the skin builds new collagen, environmental stress forces the skin to actively dismantle the dense collagen bundles it already has. The dermis effectively turns its own remodeling tools against its structural foundation.
Understanding what enzymes break down collagen fibers in the dermis requires tracing the MMP family, observing the initial cut, mapping the fragment breakdown, identifying the UV and ROS triggers, contrasting healthy remodeling with pathological degradation, and deploying exact inhibition strategies.
What is the biological baseline for how enzymes break down collagen fibers in the dermis?
The biological baseline is that Matrix Metalloproteinases normally participate in controlled extracellular-matrix turnover, tissue remodeling, and wound repair, but they become damaging when their activity rises high enough that collagen destruction outpaces collagen replacement.
In normal healing, MMP activity is tightly controlled to remove damaged tissue and make room for fresh synthesis. In pathological or photoaged skin, that careful balance is permanently lost, leaving the dermis in a persistent state of degradation.
MMPs are specialized, zinc-dependent enzymes capable of degrading almost all extracellular matrix proteins. When activated chronically, they ensure that the structural scaffolding of the skin is relentlessly degraded.
Quan et al. (2009) reported that, among the 19 MMPs expressed in normal human skin, only three were significantly induced by UV irradiation in their in-vivo experiment: MMP-1, MMP-3, and MMP-9. That makes this specific trio the clearest quantitative anchor for dermal collagen breakdown in photoaging [PMC].
| Enzyme Family Member | Primary Classification | Main Role in Degradation |
|---|---|---|
| MMP-1 | Collagenase | Initiates the first cleavage of intact fibrillar collagen. |
| MMP-3 | Stromelysin | Amplifies downstream activity and degrades broader matrix components. |
| MMP-9 | Gelatinase | Clears and further breaks down the degraded collagen remnants. |
How does MMP-1 initiate how enzymes break down collagen fibers in the dermis?
MMP-1 initiates how enzymes break down collagen fibers in the dermis by acting as the primary collagenase that cleaves fibrillar collagen, especially Type I collagen and Type III collagen, at a specific vulnerable site within the collagen triple helix.
This enzyme performs the most difficult mechanical task: breaching the rigid, intact structure of a healthy collagen fiber. Once that first critical cut occurs, the fiber loses much of its normal structural integrity and uncoils, exposing itself to secondary degradation.
Because MMP-1 is the primary initiator, an increase in its activity guarantees widespread architectural failure inside the dermal matrix.
Quan et al. (2009) state that MMP-1 initiates the cleavage of fibrillar collagen, typically type I and III in skin, at a single site within its central triple helix. This “single-site” cleavage model is the core quantitative-mechanistic explanation for how structural ruin begins [PMC].
How does the clipping mechanism explain how enzymes break down collagen fibers in the dermis?
The clipping mechanism explains dermal collagen breakdown because collagen fibers are not dissolved all at once. The enzyme first recognizes a susceptible site, binds to the collagen structure, and then acts like a molecular scissor that cuts the triple helix into weaker fragments.
Those fragments then lose mechanical tension and matrix-support value. The destruction follows a physical progression: without targeted binding and cleavage, the resilient collagen fiber would remain completely immune to ambient stress.
This collagen cleavage guarantees that the matrix becomes fragmented rather than instantly erased, causing dermal firmness to decrease stepwise as more fibers succumb to enzymatic clipping.
| Destructive Sequence | Molecular Action | Dermal Consequence |
|---|---|---|
| 1. Recognition Phase | Enzyme binds to a susceptible collagen site | Initiation of structural threat |
| 2. Cleavage Phase | Collagen helix is cut into fragments | Physical continuity broken |
| 3. Fragmentation Phase | Supportive structure weakens and uncoils | Loss of tensile strength |
| 4. Net Effect | Fragmented matrix replaces solid scaffold | Firmness and matrix cohesion decrease |
How do MMP-3 and MMP-9 continue how enzymes break down collagen fibers in the dermis?
MMP-3 and MMP-9 continue how enzymes break down collagen fibers in the dermis by processing and extending the degradation that begins after MMP-1 makes the first cut.
MMP-3 broadens matrix-degrading activity and supports widespread activation cascades. MMP-9, operating as a gelatinase, helps degrade collagen remnants and other uncoiled matrix fragments further.
They act as ruthless amplifiers, ensuring that the initial damage caused by MMP-1 is completed. By aggressively clearing the fragmented remnants, they prevent the collagen from re-establishing any functional mechanical support.
Quan et al. (2009) reported that MMP-1 and MMP-3 mRNA were induced several thousand-fold at 24 hours after UV exposure, while MMP-9 was induced about 6-fold. The review confirms that once collagen is cleaved by MMP-1, it can be further degraded by elevated MMP-3 and MMP-9 [PMC].
How does UV radiation trigger the enzymes that break down collagen fibers in the dermis?
UV radiation triggers these enzymes by activating stress-signaling pathways in the skin that drastically increase MMP expression and shift the dermis toward a highly degradative state.
In photoaging literature, UV exposure is the absolute most important external trigger for excess collagen-destructive enzyme activity. It is the upstream catalyst that initiates the entire biochemical cascade.
By exploring how UV radiation degrades dermal collagen, it becomes clear that ultraviolet light is not merely “drying out” the skin; it is actively rewriting the genetic instructions of the tissue, commanding fibroblasts to churn out massive quantities of destructive MMPs instead of healthy structural proteins.
MMP Activation Flowchart
- 1. UV rays strike the skin.
- 2. Reactive Oxygen Species are generated.
- 3. Stress-signaling pathways activate (e.g., AP-1, NF-κB).
- 4. MMP production rises exponentially.
- 5. Collagen breakdown accelerates wildly.
How does oxidative stress amplify why enzymes break down collagen fibers in the dermis?
Oxidative stress amplifies dermal collagen breakdown because Reactive Oxygen Species (ROS) increase molecular stress inside skin, strengthen pro-degradation signaling, and also make it harder for fibroblasts to maintain efficient collagen replacement.
ROS act as the vital bridge between UV exposure and enzymatic collapse. They amplify degradative signaling while simultaneously creating a toxic environment that suppresses procollagen synthesis. That creates a devastating double burden: more breakdown and far weaker repair.
This means collagen destruction rises massively while recovery falls, ensuring net matrix loss accelerates.
How does balanced remodeling compare to conditions where enzymes break down collagen fibers in the dermis excessively?
Balanced remodeling differs from excessive dermal collagen degradation because healthy skin keeps matrix turnover and collagen replacement in equilibrium, while photoaged or stressed skin shifts into net collagen loss when MMP activity is too strong or too persistent to be matched by repair.
Controlled turnover maintains dermal stability. In contrast, when enzymes break down collagen fibers in the dermis excessively, the replacement machinery is completely overwhelmed, resulting in a fragmented, weakened, and structurally compromised dermis.
| Biological State | MMP Activity | Collagen Synthesis | Net Matrix Result |
|---|---|---|---|
| Healthy Remodeling | Controlled | Keeps pace with turnover | Stable, firm dermis |
| Excessive Degradation | Overactive | Too weak to compensate | Fragmented, weakened dermis |
How does photoaging differ from intrinsic aging when enzymes break down collagen fibers in the dermis?
Photoaging differs from intrinsic aging because UV exposure actively accelerates enzymatic collagen destruction, while intrinsic aging is more strongly associated with a slower, more gradual decline in collagen renewal. Knowing why collagen density decreases in the dermis with age helps differentiate this gradual thinning from the violent tearing caused by solar radiation.
Intrinsic aging reduces renewal gradually over decades. Photoaging, however, accelerates enzyme-driven destruction radically, ensuring that sun-exposed skin ages far faster, more fragmented, and much more chaotically than tissue experiencing normal chronological decay.
What specific skincare ingredients inhibit the enzymes that break down collagen fibers in the dermis?
The most useful skincare strategies inhibit these enzymes indirectly by blocking the UV trigger, reducing ROS-driven signaling, or supporting collagen-related repair pathways. In practical terms, the strongest long-term categories are broad-spectrum sunscreen, antioxidants, and topical retinoids.
Trigger blocking plus oxidative-stress reduction plus repair-support signaling ensures that excessive collagen degradation slows dramatically, vastly improving long-term dermal preservation.
How does broad-spectrum sunscreen inhibit the enzymes that break down collagen fibers in the dermis?
Broad-spectrum sunscreen helps inhibit this enzymatic process upstream because it reduces the UV exposure that initiates oxidative signaling and MMP overactivation. It is the absolute most important preventive step in limiting UV-driven collagen damage.
By providing rigorous UV blocking, sunscreen creates an environment with less upstream MMP and ROS activation, guaranteeing that collagen degradation slows before it even has the chance to start.
Hughes et al. (2013) found in a randomized trial that daily sunscreen use led to 24% less skin aging over 4.5 years compared with discretionary use. This is one of the strongest clinical anchors for the preventive role of sunscreen in photoaging-related collagen preservation [PubMed].
How do antioxidants inhibit the enzymes that break down collagen fibers in the dermis?
Antioxidants help inhibit these enzymes indirectly by lowering the ROS burden that feeds upstream stress signaling. Vitamin C, vitamin E, and green tea polyphenols are the most useful examples to frame as supportive anti-photoaging ingredients.
By providing a lower oxidative-stress burden, less upstream enzyme-activating signaling occurs, meaning dermal collagen breakdown is far less aggressively amplified.
How do topical retinoids inhibit the enzymes that break down collagen fibers in the dermis?
Topical retinoids help inhibit excess collagen degradation by improving collagen-related signaling, partially countering matrix disruption, and supporting better long-term renewal behavior in photodamaged skin. Their role is highly supportive and restorative rather than instant.
Tretinoin signaling improves collagen-supportive behavior, ensuring the matrix balance shifts away from pure degradation so that photodamaged skin becomes more repair-capable over time.
Griffiths et al. (1993) reported that collagen I formation was 56% lower in photodamaged skin than in sun-protected skin, and that topical tretinoin produced an 80% increase in collagen I formation, whereas vehicle treatment showed a 14% decrease. This is one of the clearest numeric anchors for retinoid support of collagen-related recovery behavior [PubMed].
Anti-Degradation Intervention Pathway
- Problem → MMP activity is accelerated by UV and oxidative stress.
- Implication → collagen fibers are cut faster than they are replaced.
- Solution → block UV, reduce ROS, and improve collagen-supportive signaling.
What are the key summary facts about the enzymes that break down collagen fibers in the dermis?
The key summary facts are that the main enzymes are Matrix Metalloproteinases, especially MMP-1, and that MMP-3 and MMP-9 help continue the degradation after the first cut. UV and oxidative stress are the biggest external triggers of excessive activity, and prevention works best when the trigger, amplification, and replacement imbalance are all addressed together.
Summary Checklist
What steps can you take to control the enzymes that break down collagen fibers in the dermis today?
Daily control works best when dermal collagen degradation is treated as a cumulative biochemical process that needs steady interruption, not a one-time correction. The practical priorities are daily broad-spectrum sunscreen, antioxidant support where appropriate, cautious retinoid use if tolerated, and lower cumulative UV exposure overall.
Final Execution Checklist
Quick Answers About the Enzymes That Break Down Collagen Fibers in the Dermis
What enzymes break down collagen fibers in the dermis?
The main enzymes are Matrix Metalloproteinases, especially MMP-1, which starts collagen cleavage. MMP-3 and MMP-9 help continue the breakdown after the first cut, turning damaged collagen into smaller unusable fragments.
What does MMP-1 do to collagen?
MMP-1 acts as the key collagenase in skin photoaging. It makes the first critical cut in Type I and Type III collagen, weakening the collagen triple helix and making the fiber much easier for other enzymes to degrade further.
Do MMP-3 and MMP-9 cut collagen too?
They mainly continue the damage after MMP-1 begins it. MMP-3 helps amplify degradative activity and supports downstream pathways, while MMP-9 helps process and clear broken matrix remnants further.
Is collagen loss only about making less collagen with age?
No. Collagen loss is also an active degradation process. In photoaging, the skin not only produces collagen less efficiently, but also destroys existing collagen faster through MMP overactivity and ROS-amplified signaling.
How does UV activate collagen-degrading enzymes?
UV exposure increases oxidative stress and activates signaling pathways that raise MMP expression. That shifts skin toward a more degradative state, so collagen breakdown accelerates beyond normal remodeling.
What is the difference between healthy remodeling and excessive degradation?
Healthy remodeling is balanced: MMP activity removes damaged material while collagen replacement keeps pace. Excessive degradation happens when enzyme activity is overactive and repair cannot compensate, so the dermis loses structure over time.
Can sunscreen reduce collagen breakdown?
Yes. Sunscreen reduces the UV exposure that triggers MMP overactivation upstream. A randomized trial found 24% less skin aging over 4.5 years with daily sunscreen use compared with discretionary use, supporting its long-term preventive role.
Do retinoids really help photodamaged collagen?
They can help support collagen-related repair behavior. In a classic study, photodamaged skin had much lower collagen formation than protected skin, and topical tretinoin increased collagen I formation substantially compared with vehicle.
Do antioxidants replace sunscreen for collagen protection?
No. Antioxidants such as vitamin C, vitamin E, and green tea polyphenols can help reduce oxidative-stress signaling, but they are adjuncts, not substitutes, for UV protection.
Why do wrinkles and sagging accelerate in sun-exposed skin?
Because photoaged skin undergoes faster enzyme-driven collagen fragmentation, stronger ROS signaling, and weaker replacement by fibroblasts. Over time, that produces a fragmented dermal matrix with less support and resilience.
Conclusion
In conclusion, enzymes break down collagen fibers in the dermis through a real biochemical destruction system centered on MMP-1–initiated cleavage, followed by MMP-3/MMP-9–supported fragment breakdown, and amplified by UV-triggered oxidative stress. That is why photoaging is not just slower collagen production; it is also active matrix dismantling, which is best reduced by consistent UV protection, oxidative-stress control, and long-term collagen-supportive care.
MMP-driven cleavage combined with UV/ROS amplification and weaker replacement guarantees that net dermal collagen loss will rise unless aggressively countered. True anti-aging strategy means prevention must relentlessly target both the upstream triggers and the downstream biology to stop the enzymatic scissors before they begin to cut.